Key Features & Benefits
Broad model portfolio – DSS (UC-like), TNBS (CD-like), and OXA models covering acute, chronic, and different immune mechanisms (Th1/Th17 for TNBS, Th9 for OXA).
Multiple species/strains – C57BL/6, BALB/c mice and SD rats available.
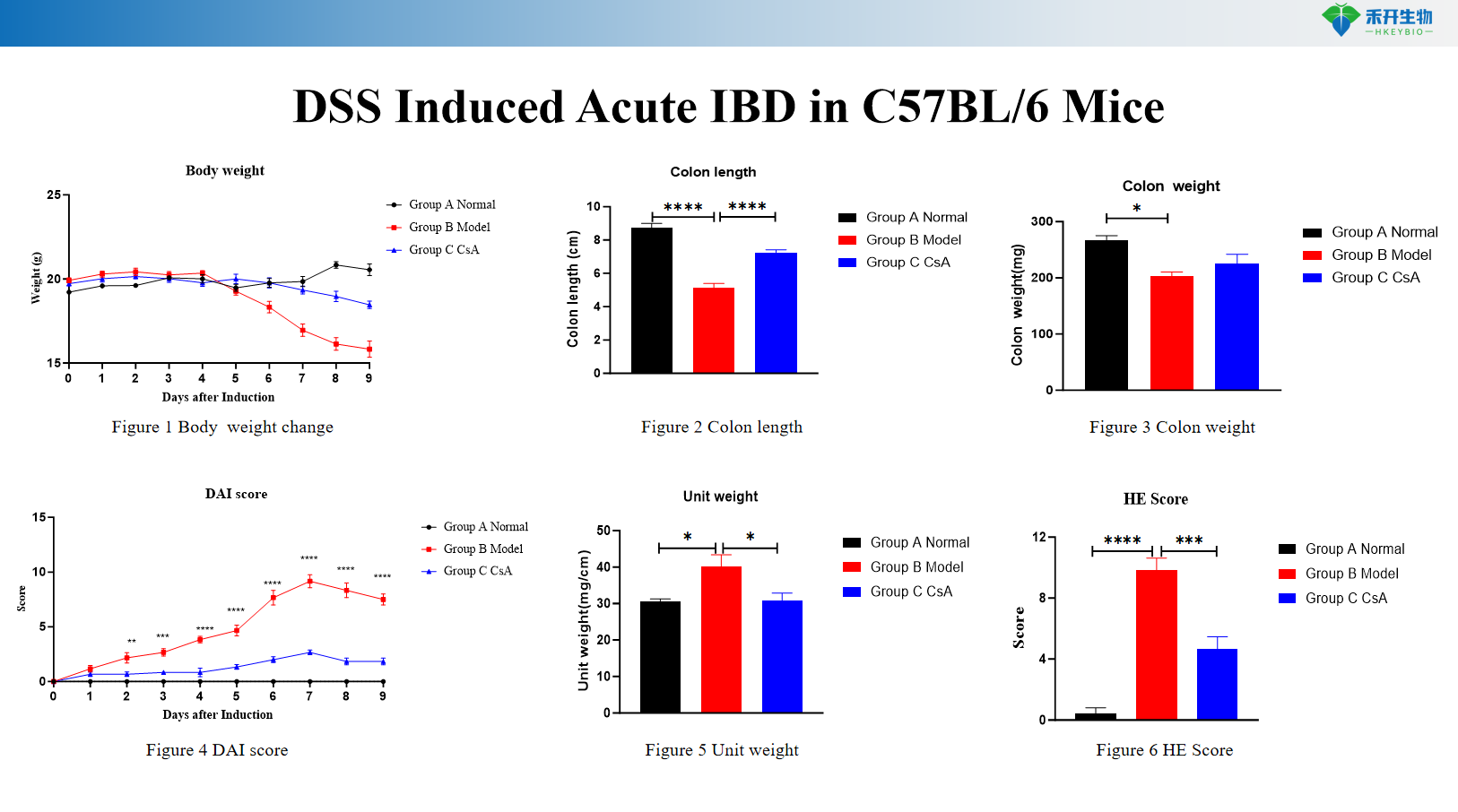
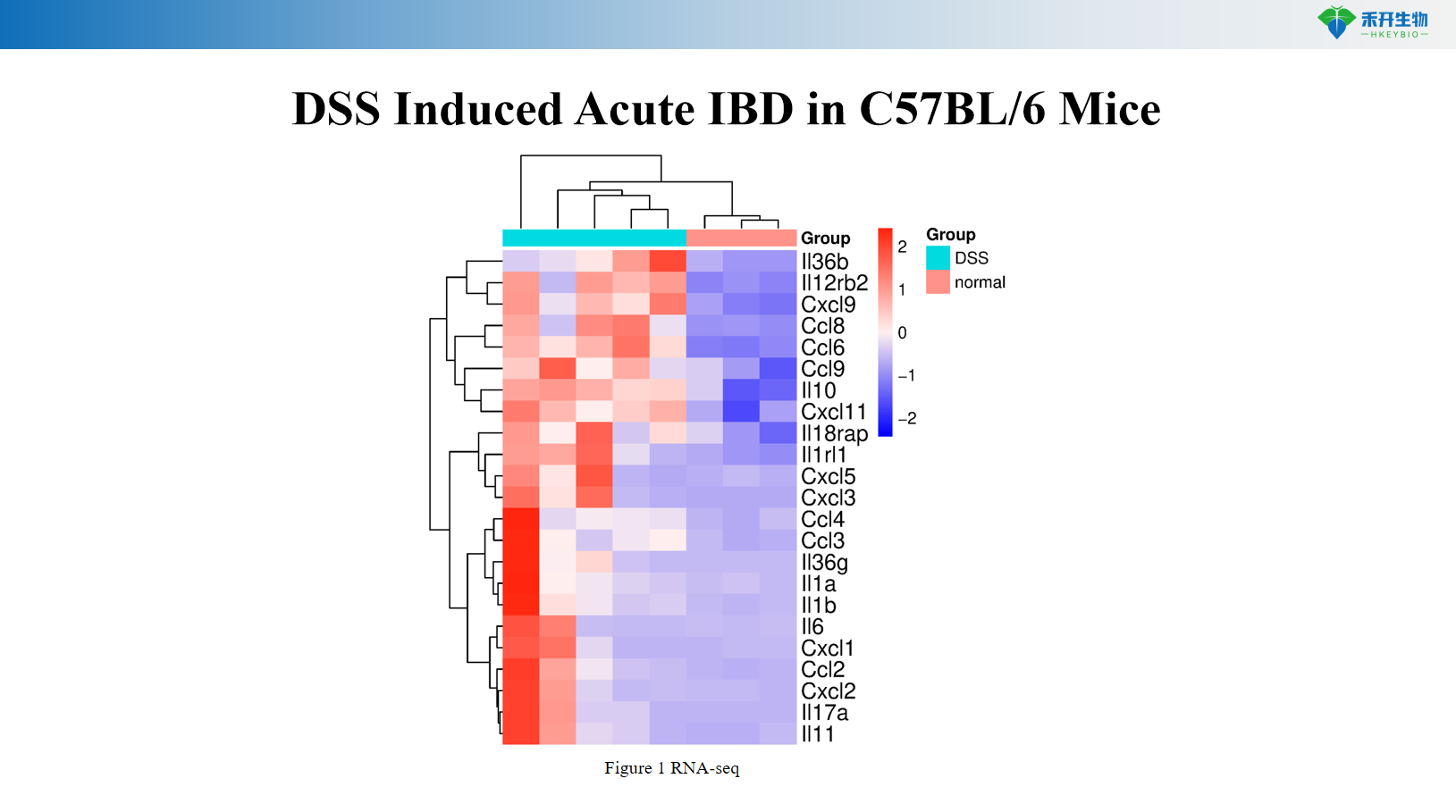
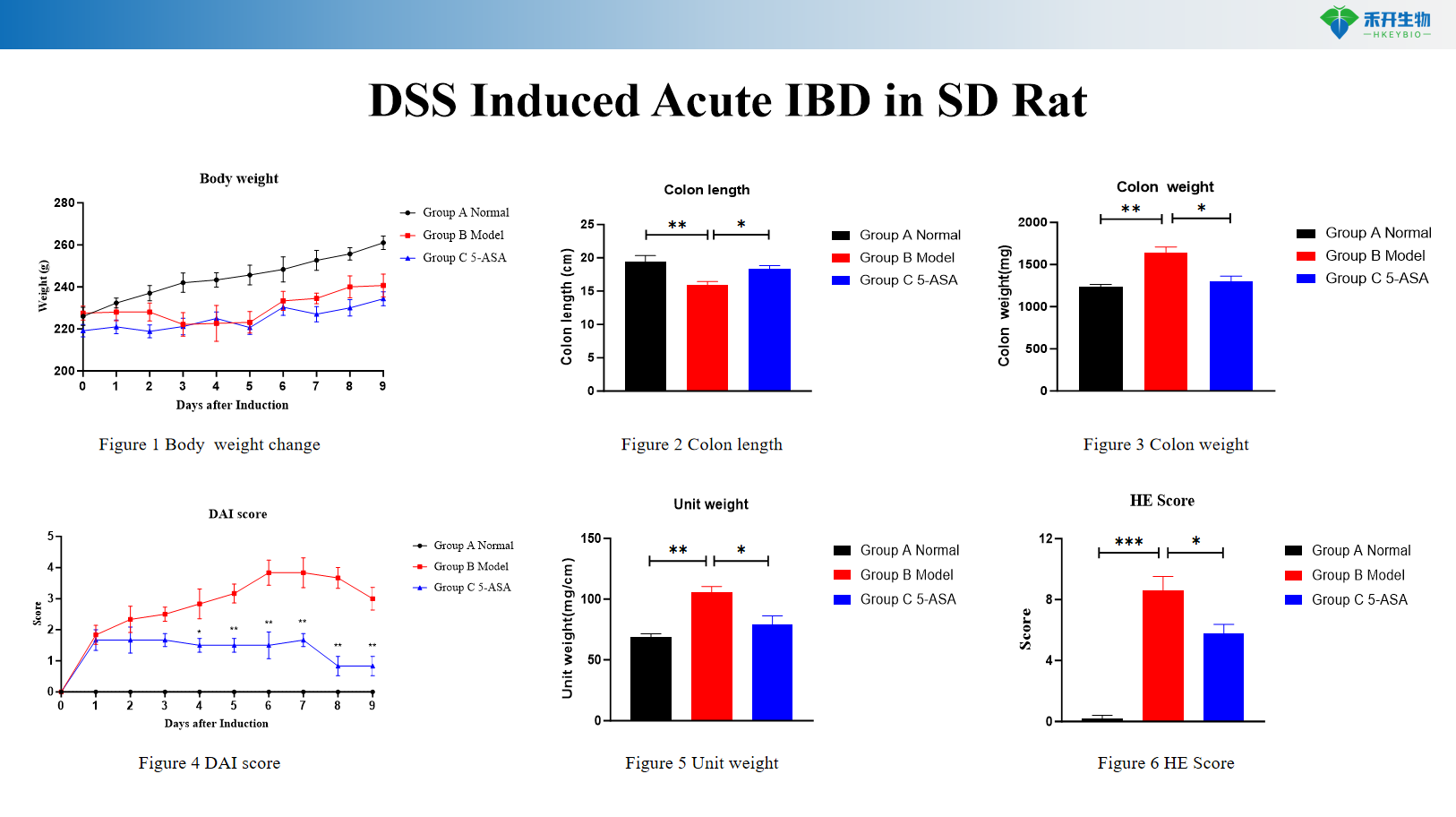
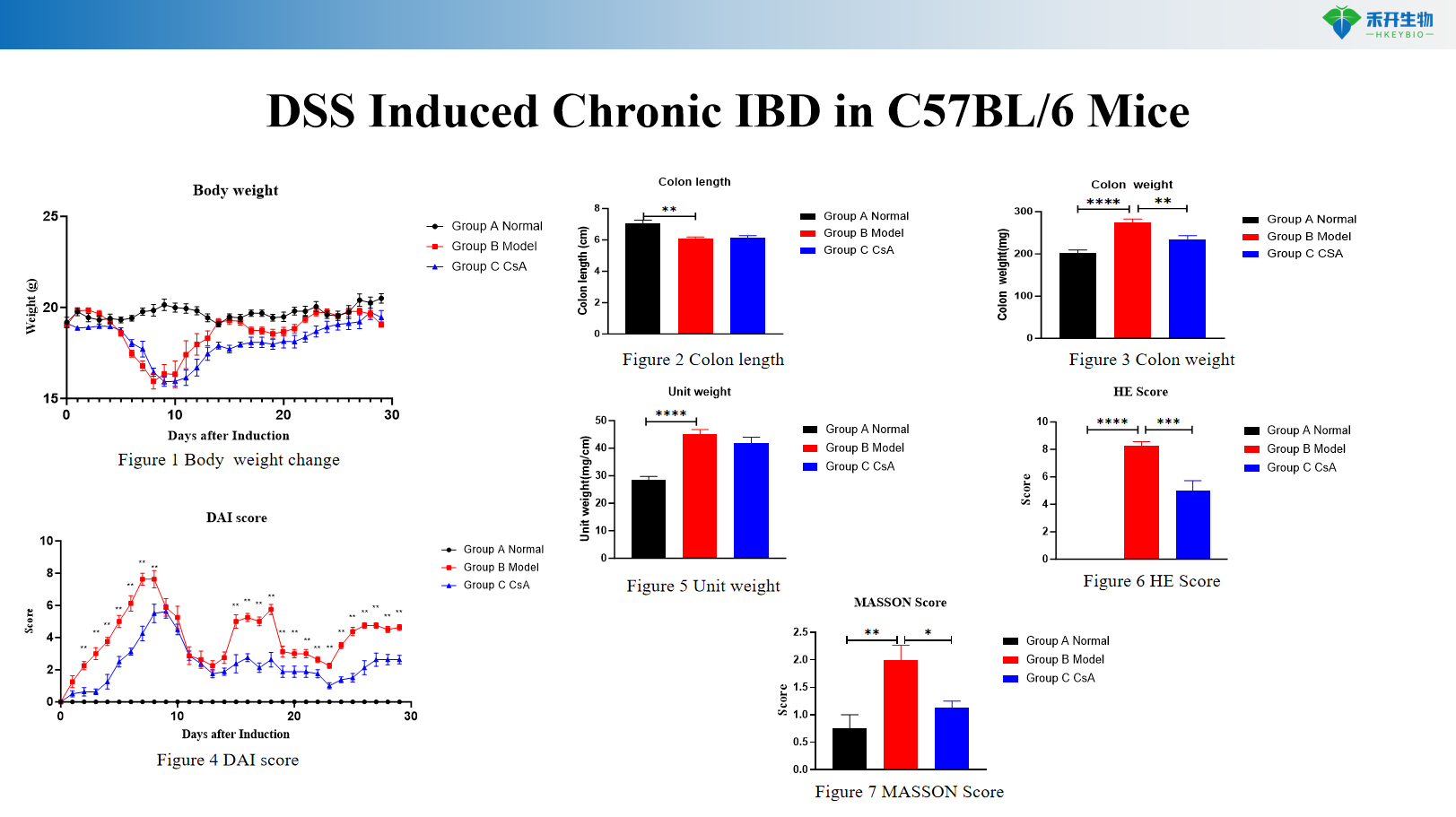
Comprehensive endpoints – Body weight, colon length/weight, DAI score, histopathology (HE, Masson), cytokine analysis (IL-6, TNF-α), RNA-seq data (chronic DSS).
Translational value – Ideal for testing anti-inflammatory drugs, biologics (anti-TNF, anti-IL-12/23), JAK inhibitors, and gut-restricted therapies.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
DSS Induced Acute IBD in C57BL/6 Mice
DSS Induced Acute IBD in SD Rat
DSS Induced Chronic IBD in C57BL/6 Mice
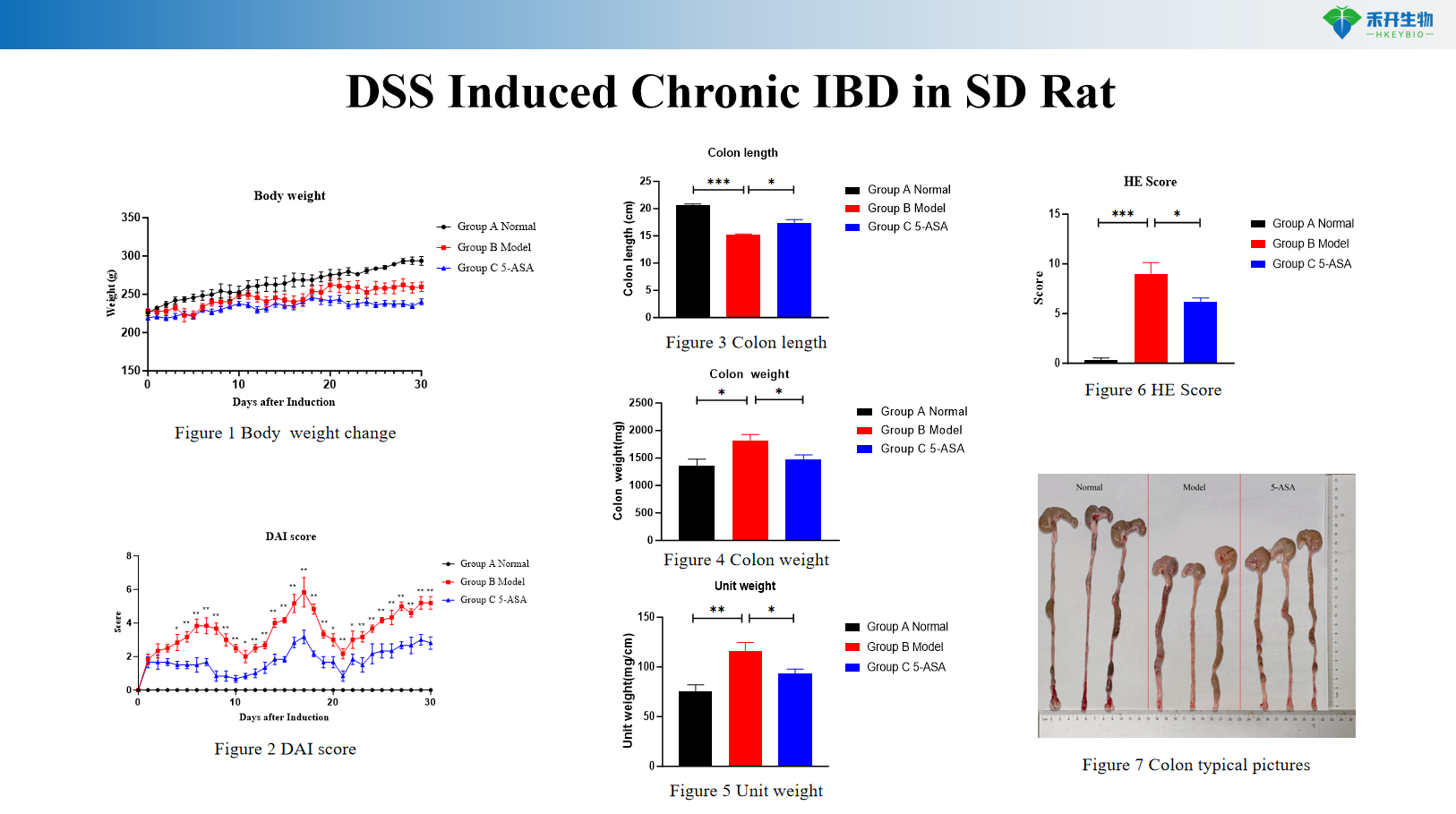
DSS Induced Chronic IBD in SD Rat
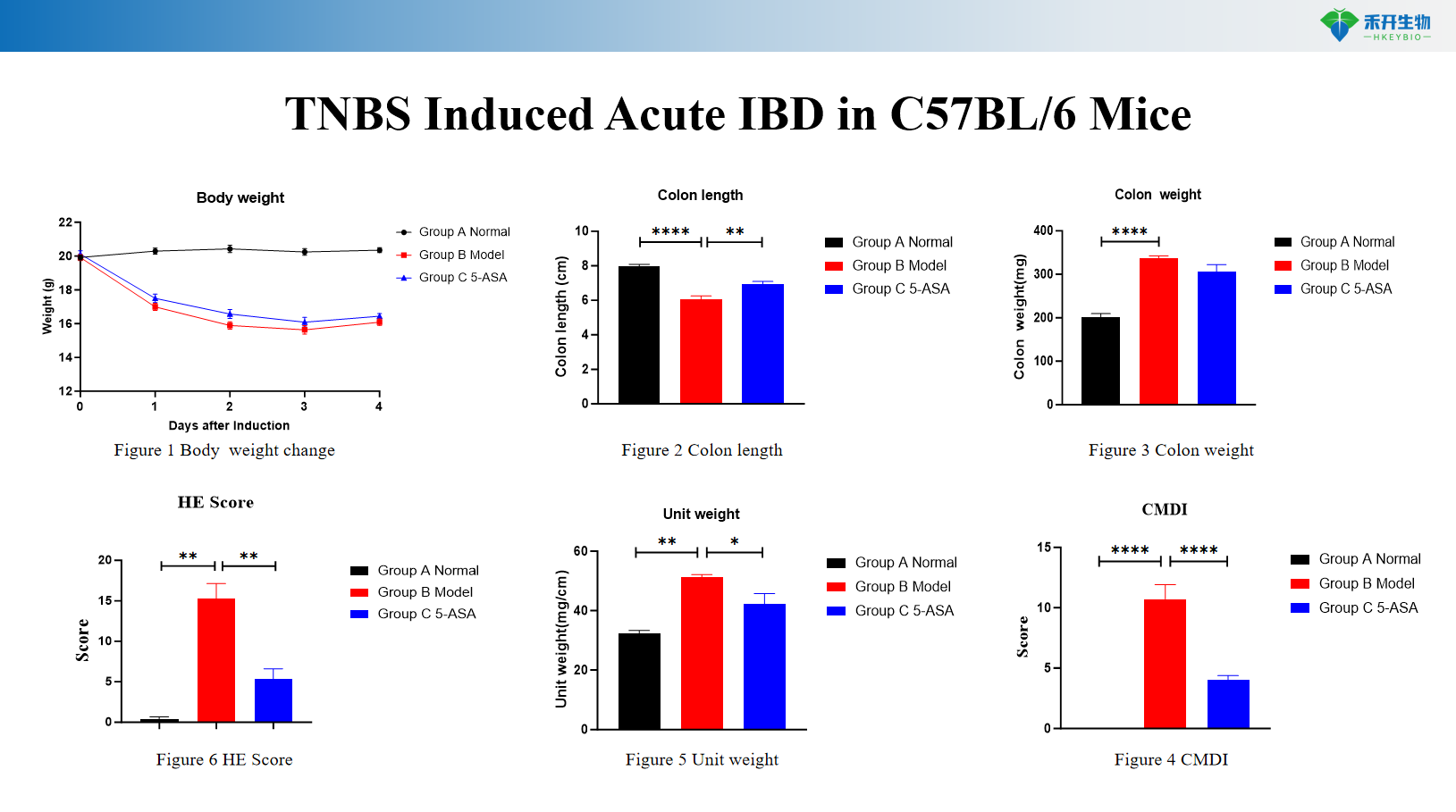
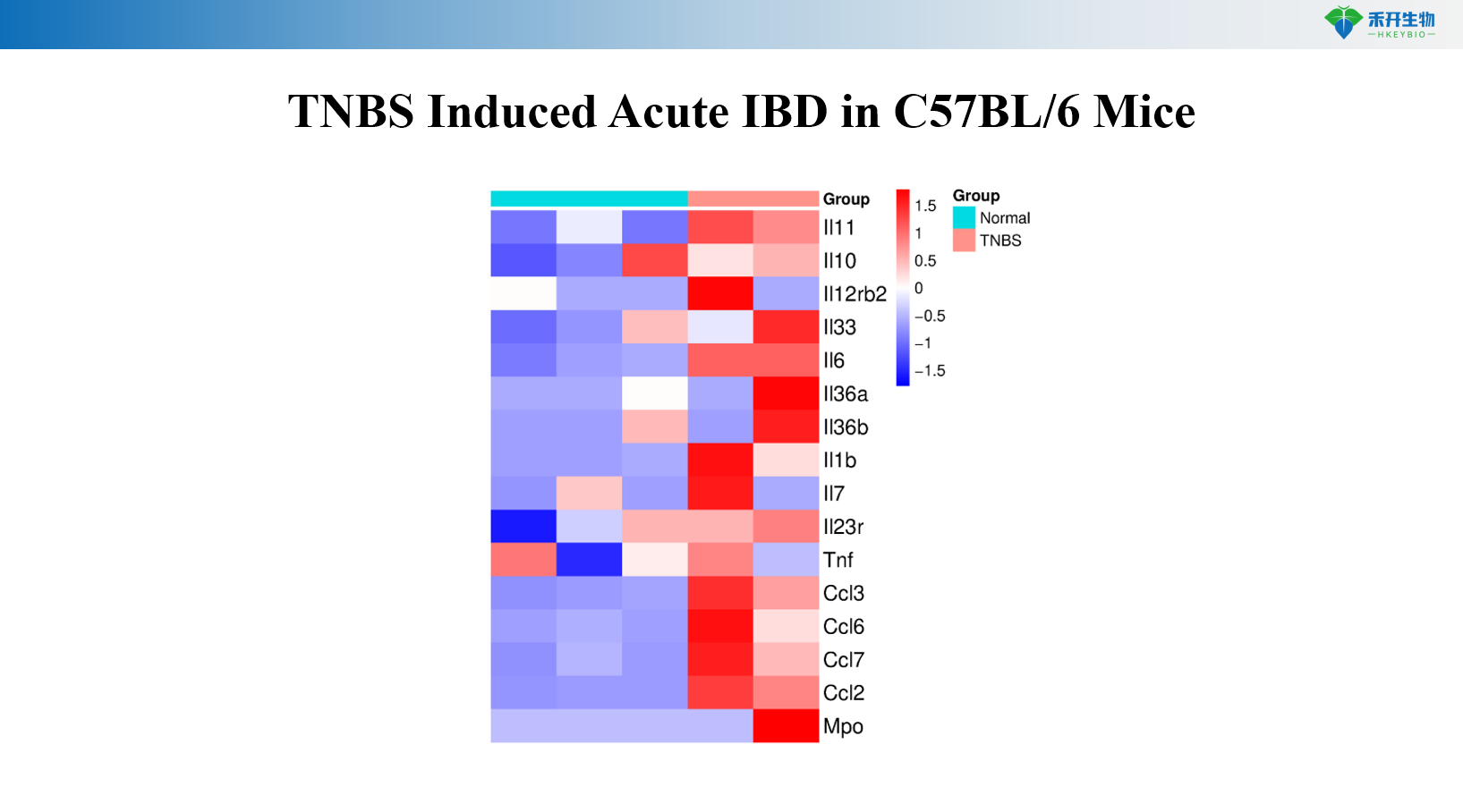
TNBS Induced Acute IBD in C57BL/6 Mice
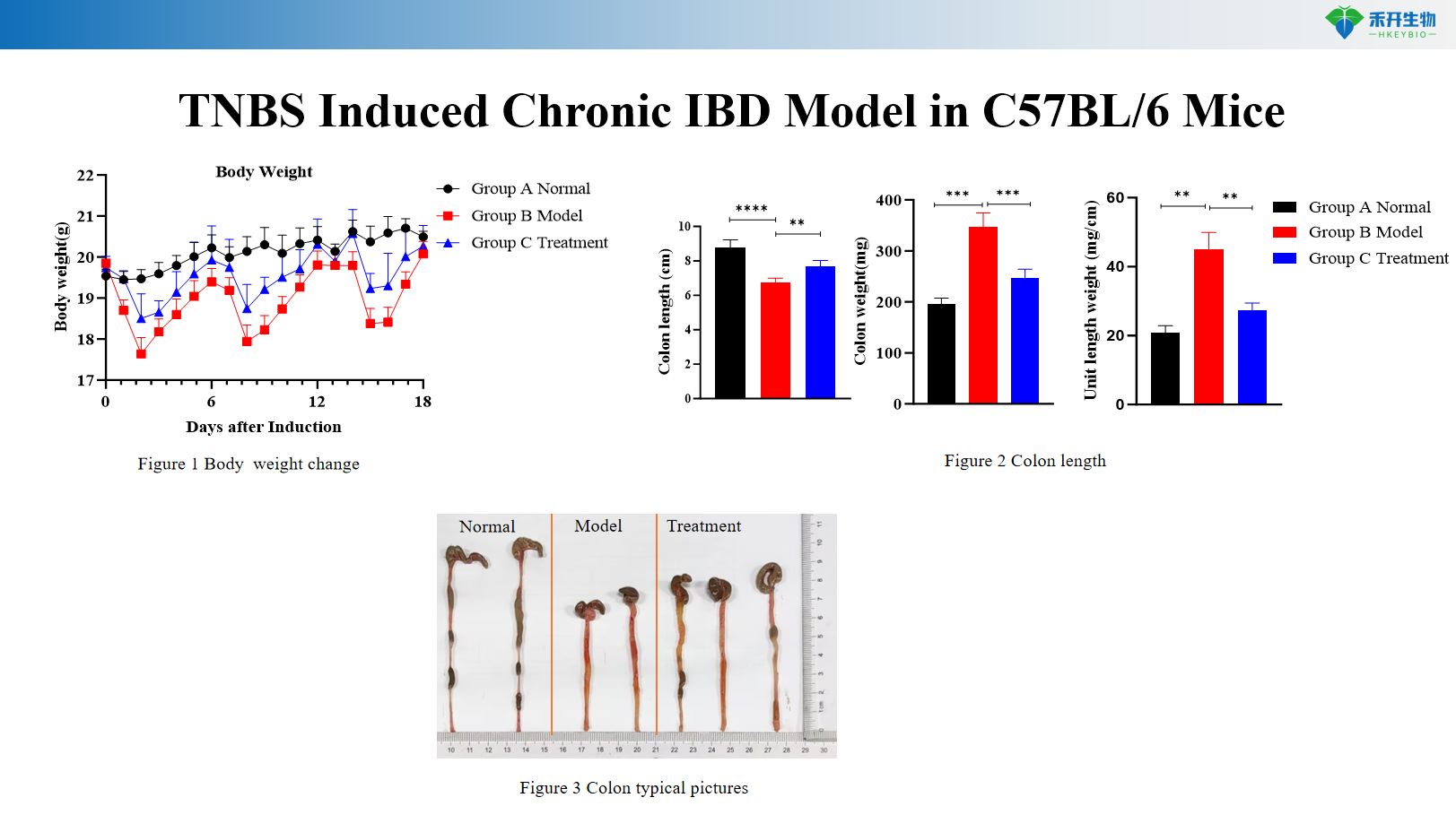
TNBS Induced Chronic IBD Model in C57BL/6 Mice
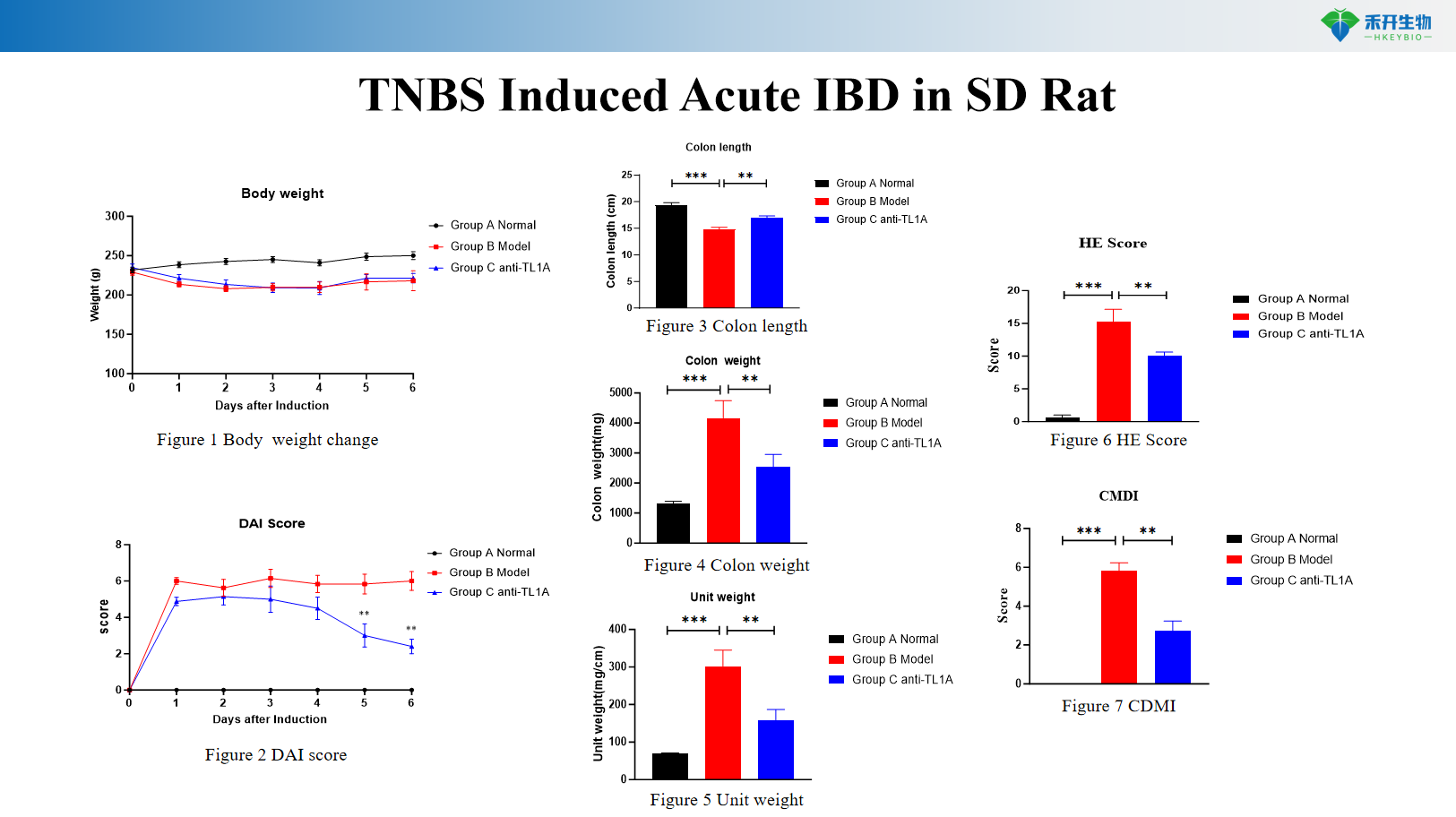
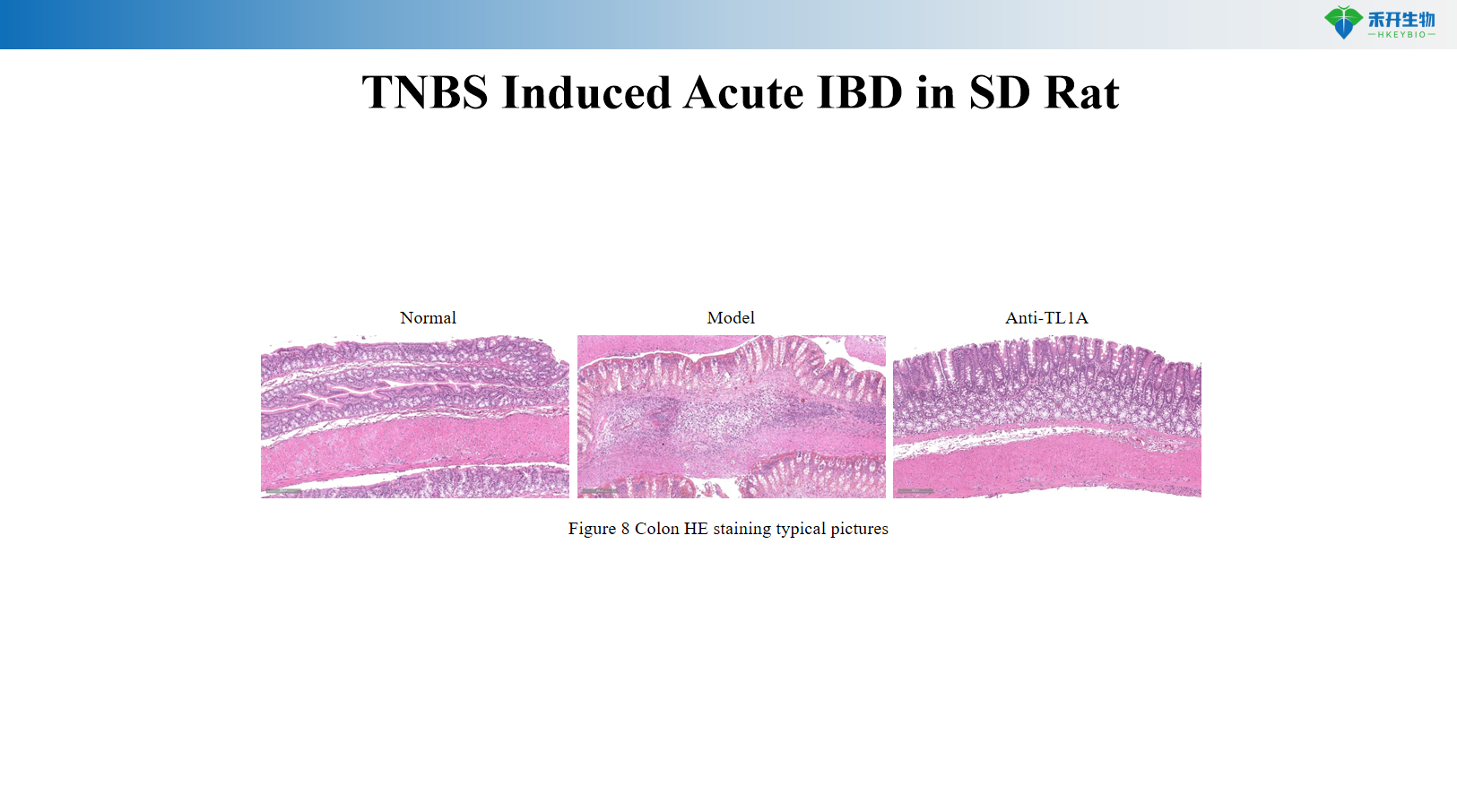
TNBS Induced Acute IBD in SD Rat
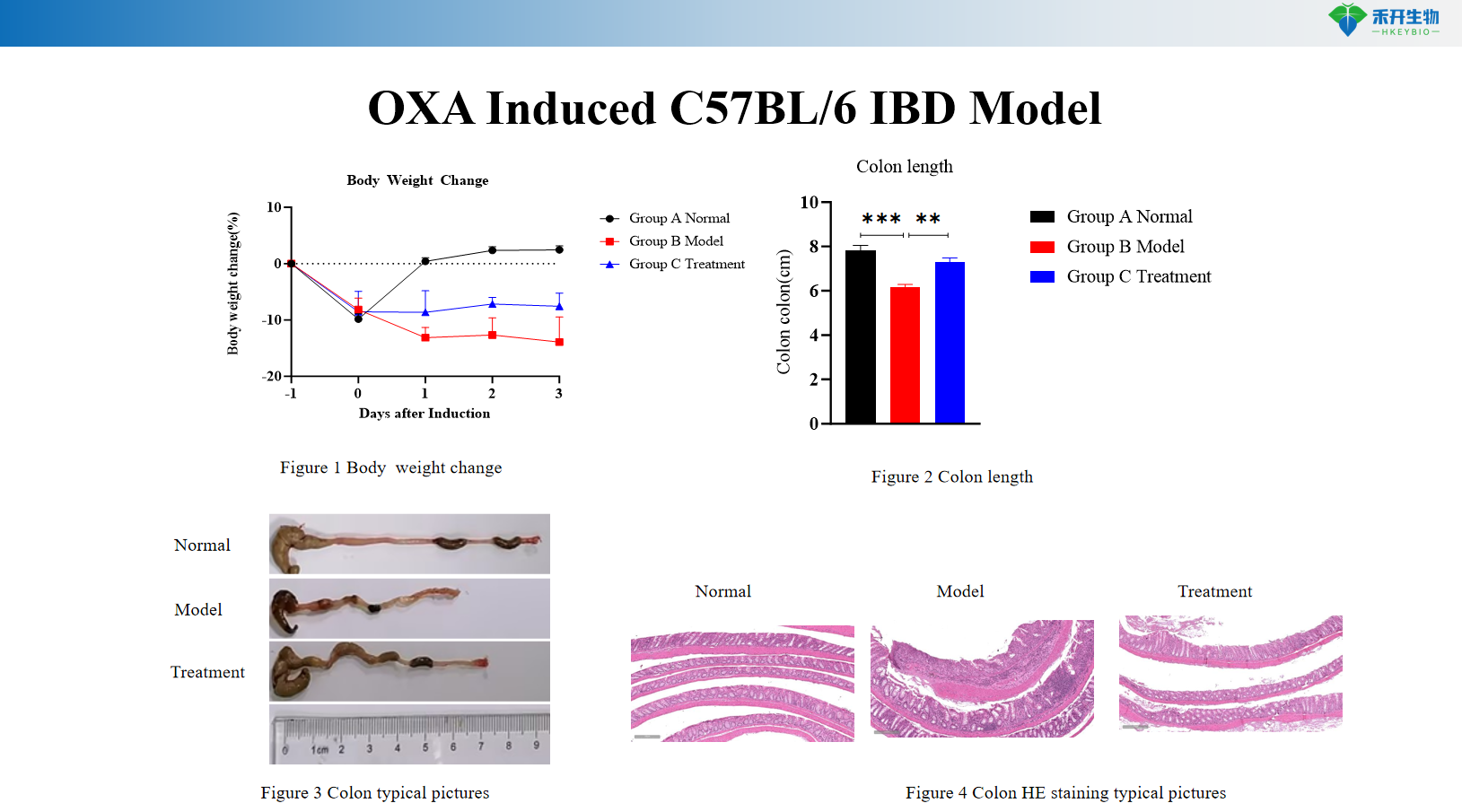
OXA Induced C57BL/6 IBD Model
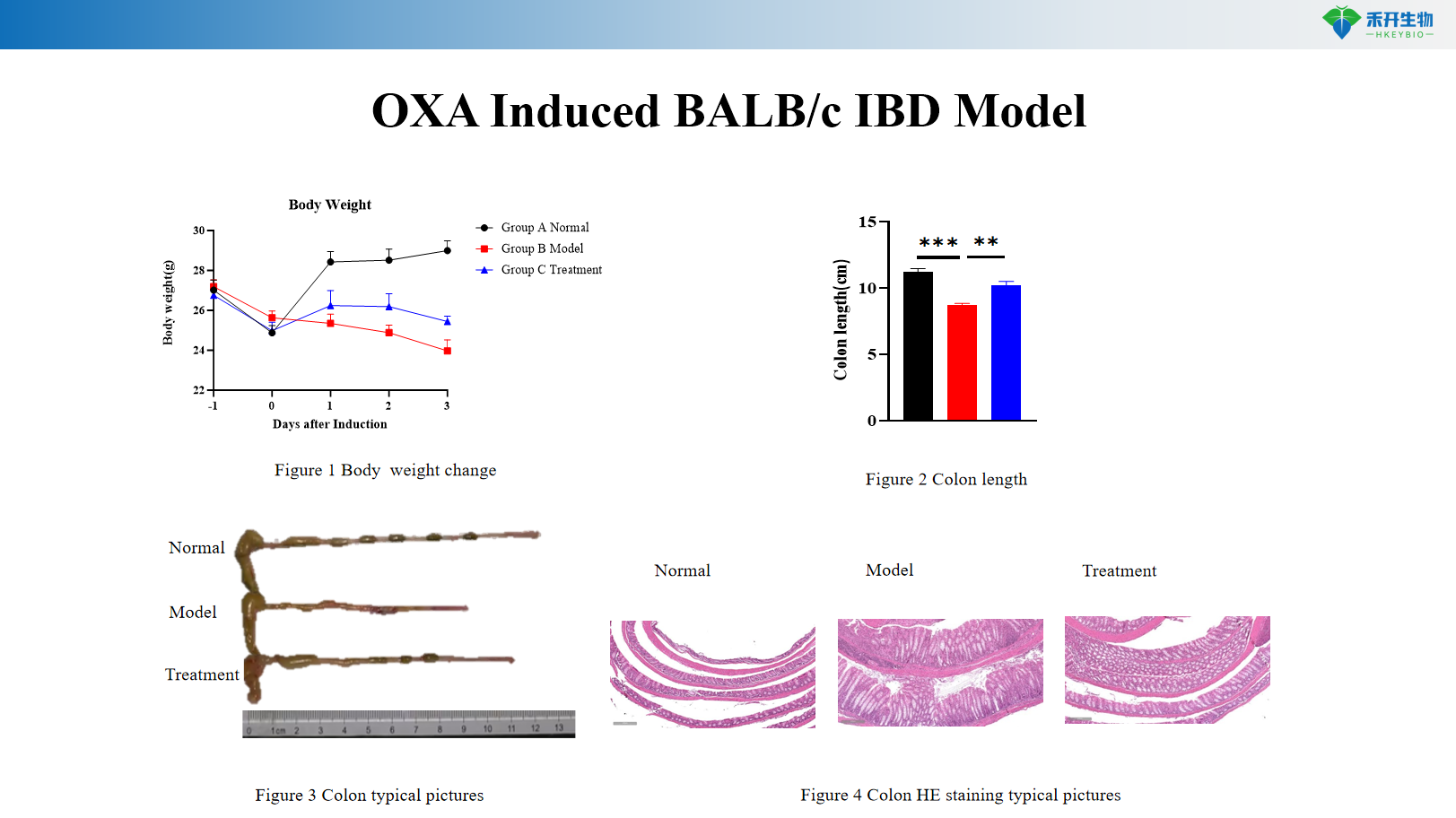
OXA Induced BALB/c IBD Model
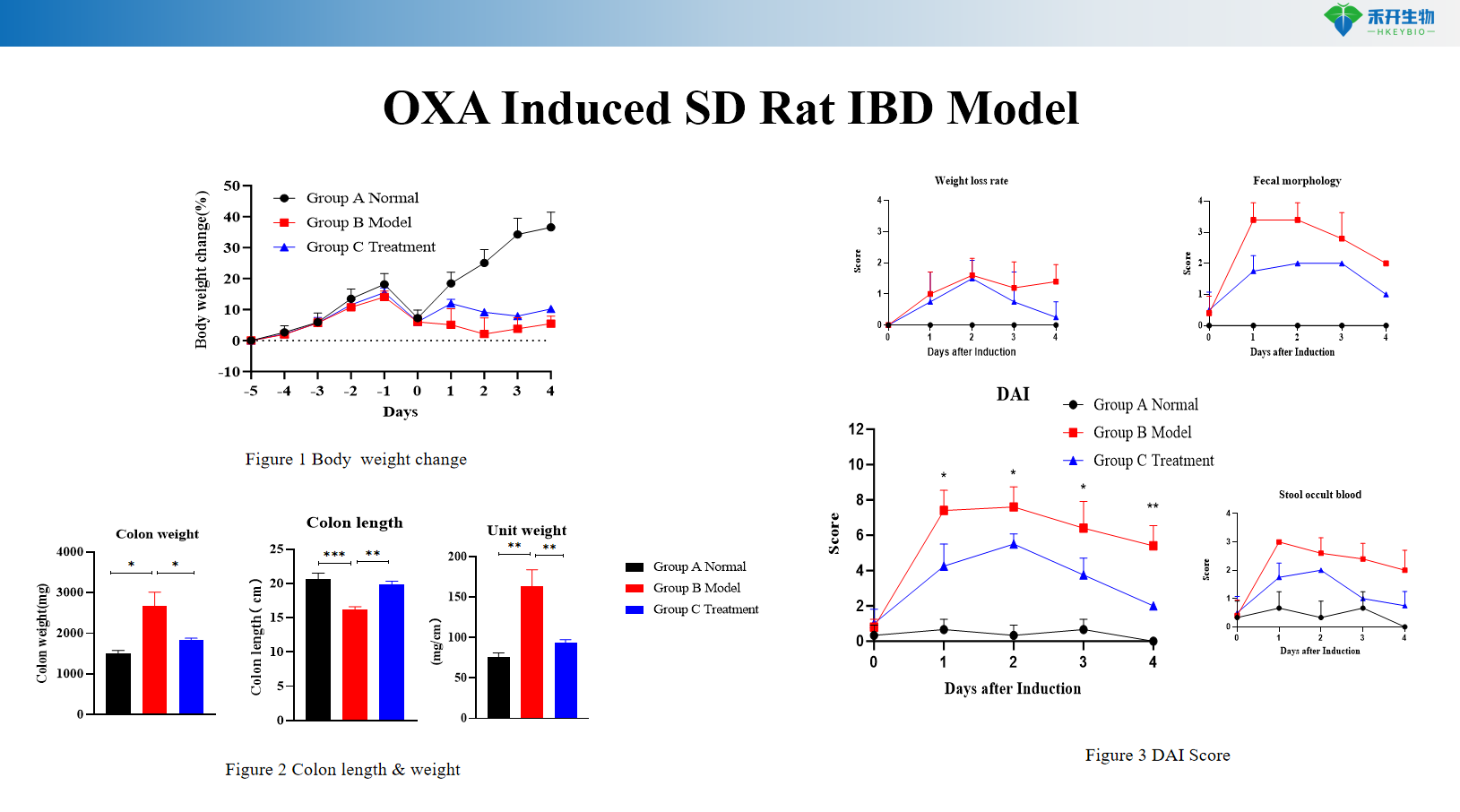
OXA Induced SD Rat IBD Model
Applications
• Efficacy testing of anti-inflammatory drugs (5-ASA, corticosteroids), biologics (anti-TNF, anti-IL-12/23, anti-integrin), JAK inhibitors, and S1P receptor modulators
• Evaluation of gut-restricted immunomodulators and microbiome-based therapies
• Target validation for Th1, Th17, Th9 pathways in IBD pathogenesis
• Biomarker discovery (fecal calprotectin, cytokines, inflammatory mediators)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strains | C57BL/6 mouse, BALB/c mouse, SD rat |
Induction methods | DSS (oral, acute 5–7 days, chronic 2–3 cycles); TNBS (intracolonic, single or repeated); OXA (intracolonic) |
Study duration | Acute: 5–10 days; Chronic: 4–8 weeks |
Key endpoints | Body weight, colon length, colon weight, DAI score (0–12 scale), histopathology (HE, Masson) with scoring, cytokine levels (IL-6, TNF-α), CMDI score (TNBS), RNA-seq data (chronic DSS), colon typical pictures |
| Positive control | 5-aminosalicylic acid (5-ASA), dexamethasone, or anti-TNF antibody available as reference compounds |
| Data package | Raw data, analysis reports, clinical scores, histology slides, ELISA results, RNA-seq data (optional), bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What are the differences between DSS, TNBS, and OXA models?
A: DSS induces ulcerative colitis-like pathology via epithelial barrier disruption. TNBS induces Crohn's disease-like transmural inflammation via Th1/Th17 responses. OXA induces Th9-mediated colitis, useful for studying distinct immune mechanisms.
Q: Which model is best for chronic IBD studies?
A: Cyclic DSS administration (2–3 cycles) produces chronic colitis with fibrosis. Repeated TNBS administration also generates chronic inflammation suitable for long-term efficacy studies.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different DSS concentrations, TNBS doses)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Acute studies: 5–10 days; chronic studies: 4–8 weeks, depending on the number of cycles and endpoints.




































