Model Portfolio Overview
Our skin inflammation animal models are organized across three major induction platforms, each targeting distinct immunological mechanisms relevant to human psoriatic disease:
1. IMQ-Driven TLR7/8 Pathway Models– Imiquimod-based induction in C57BL/6, BALB/c, and Wistar strains, ideal for studying innate immune activation and acute plaque-like pathology.
2. IL-23-Centric Th17 Activation Models – Cytokine-driven models in C57BL/6 mice and SD rats, specifically targeting the IL-23/IL-17 axis central to chronic psoriasis.
3. Combination & Co-Induction Systems – IMQ plus IL-23 or IL-23 plus IL-36 dual-pathway approaches for enhanced disease severity and complex mechanistic studies.
Key Features & Benefits
Broad psoriasis model portfolio – IMQ (TLR7/8 agonist), IL-23 (direct Th17 activation), and combination psoriasis animal models covering different immunological pathways.
Multiple species/strains – C57BL/6, BALB/c mice and Wistar rats available, enabling cross-strain validation and species-specific pharmacology assessment.
Comprehensive endpoints – PASI score, ear thickness, spleen index, body weight, IL-17A levels, skin histopathology (HE), plus optional immunohistochemistry and flow cytometry for deeper mechanistic insights.
Translational value – Ideal for testing biologics (anti-IL-17, anti-IL-23), JAK inhibitors, PDE4 inhibitors, and emerging topical therapies in a physiologically relevant context.
IND-ready data packages – Studies conducted in accordance with GLP principles, delivering regulatory-compliant documentation for FDA and EMA submissions.
Technical Data & Validation
Each psoriasis animal model in our portfolio has been rigorously validated through extensive internal characterization and peer-reviewed publications. The following sections provide detailed technical descriptions of each preclinical psoriasis model, including induction methodology, pathological hallmarks, translational applications, and representative validation data. These psoriasis mouse models and rat platforms are maintained under stringent barrier conditions with standardized protocols to ensure reproducibility across studies and sites.
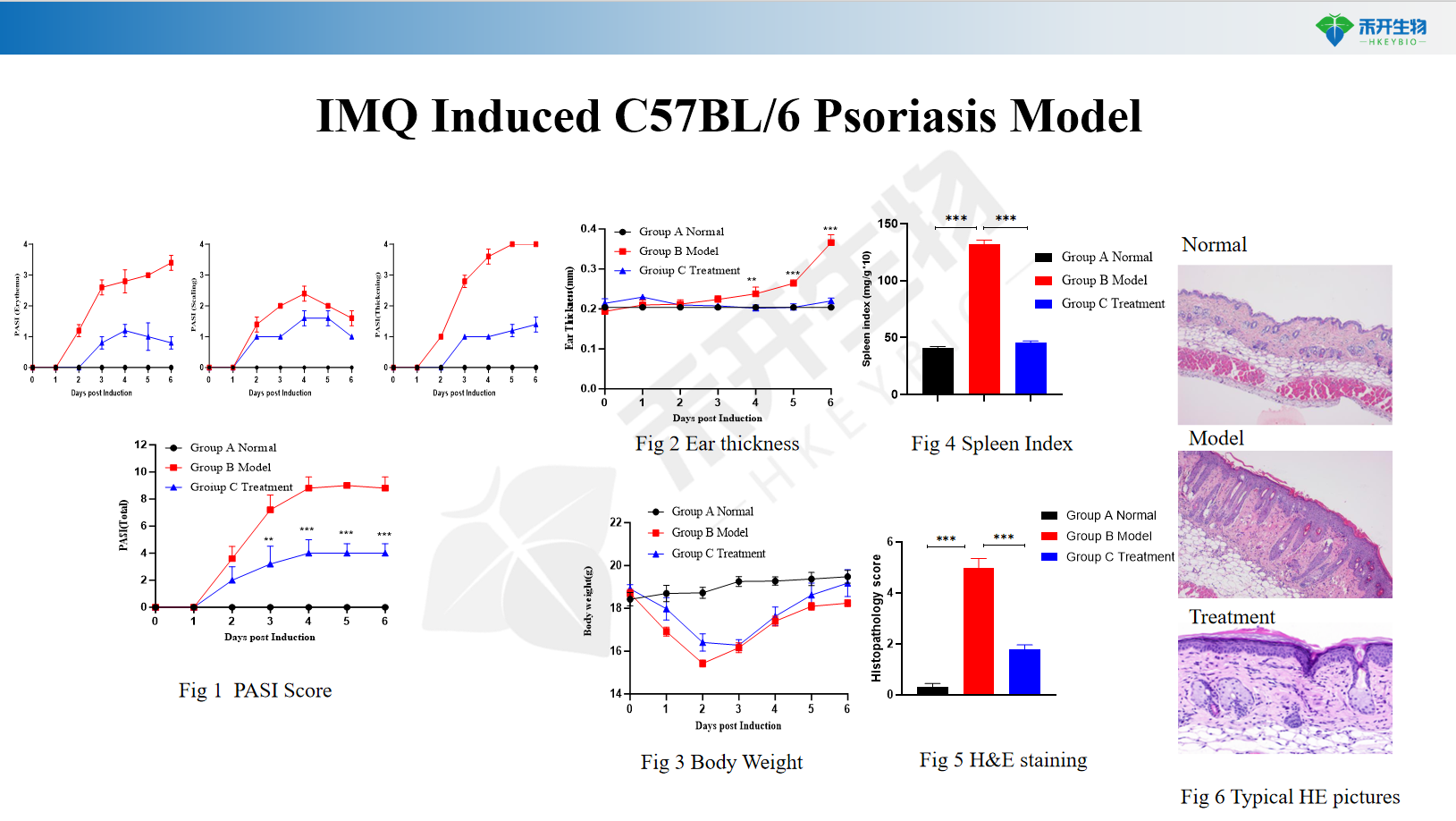
IMQ Induced C57BL/6 Psoriasis Model
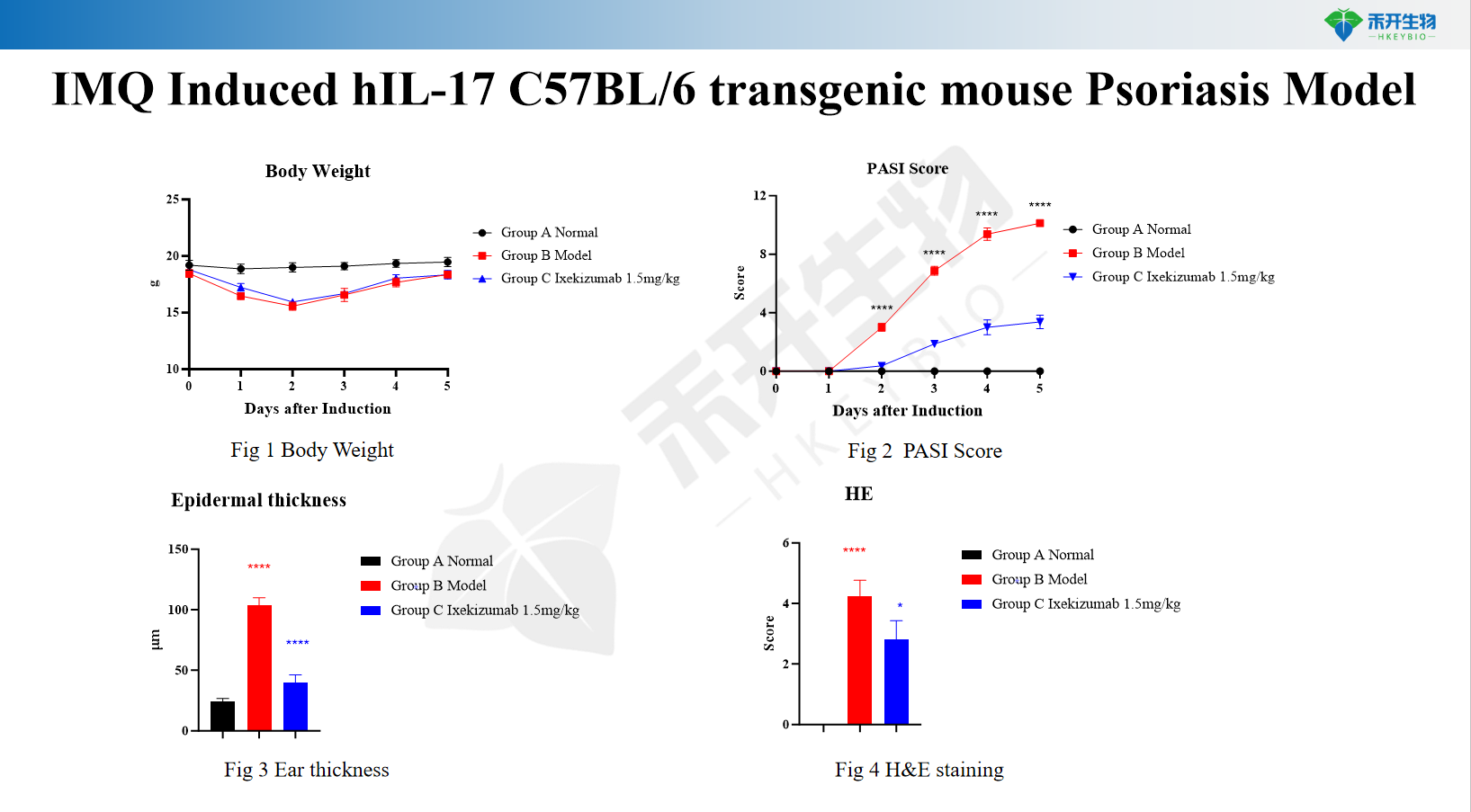
IMQ Induced hIL-17 C57BL/6 transgenic mouse Psoriasis Model
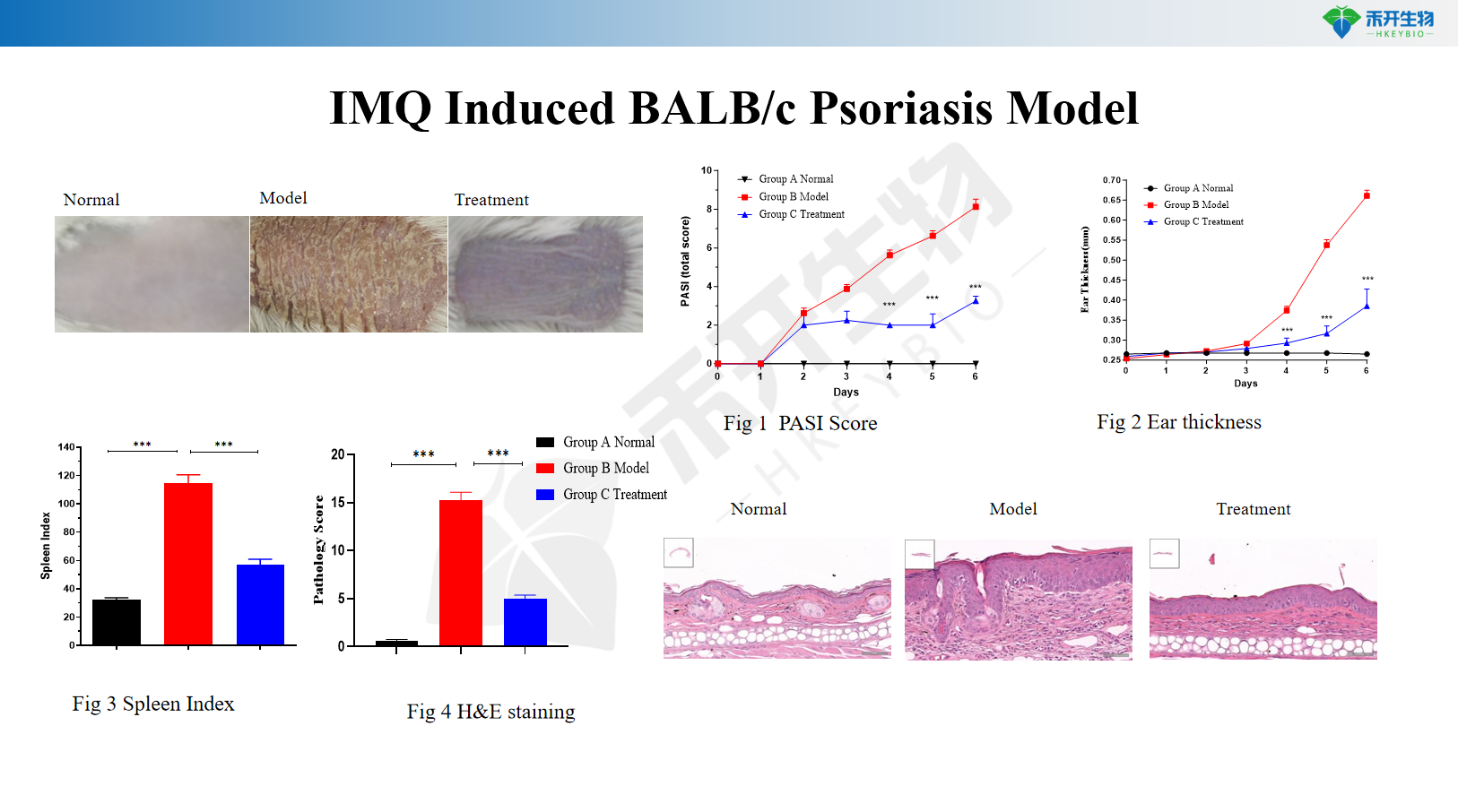
IMQ Induced BALB/c Psoriasis Model
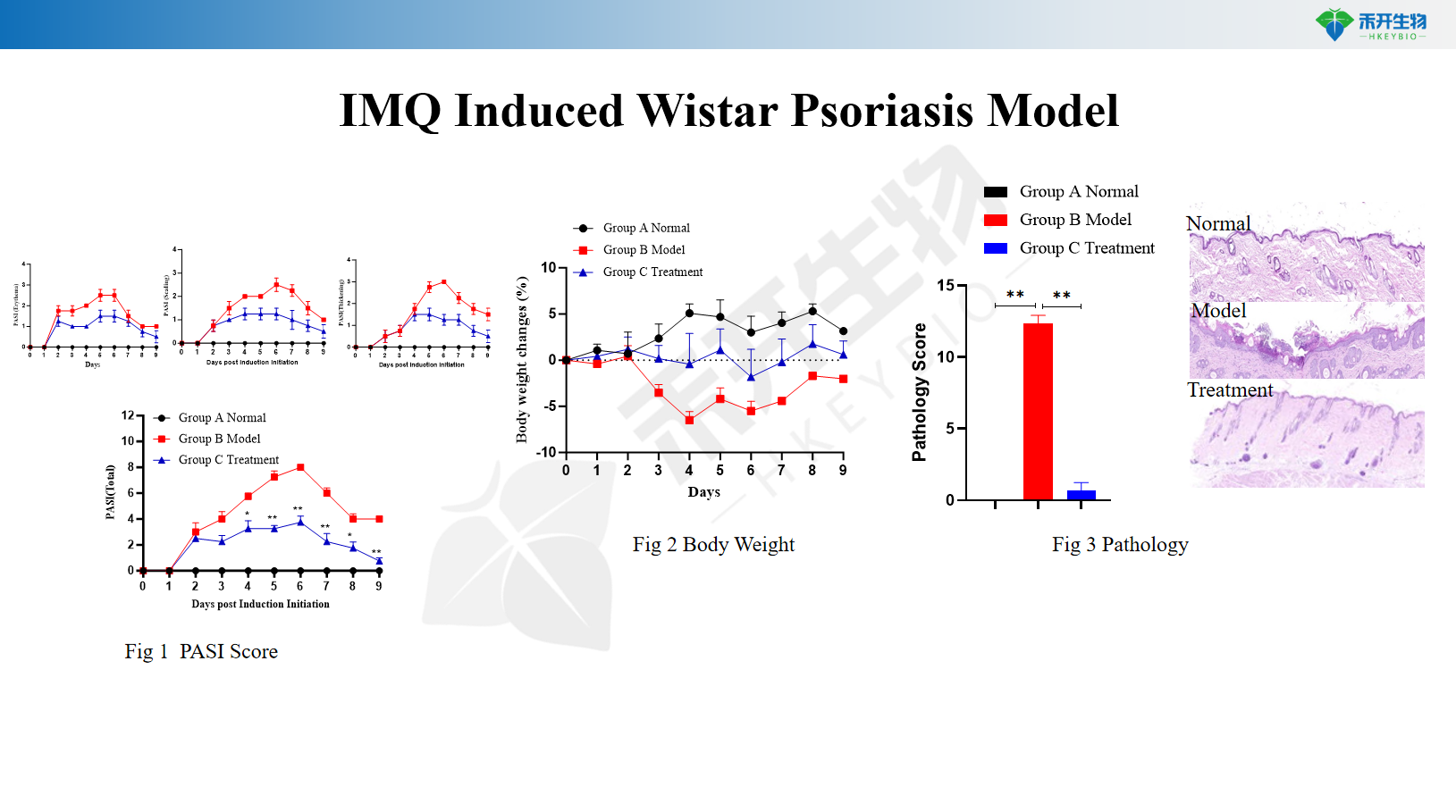
IMQ Induced Wistar Psoriasis Model
IL-23 Induced C57BL/6 Psoriasis Model
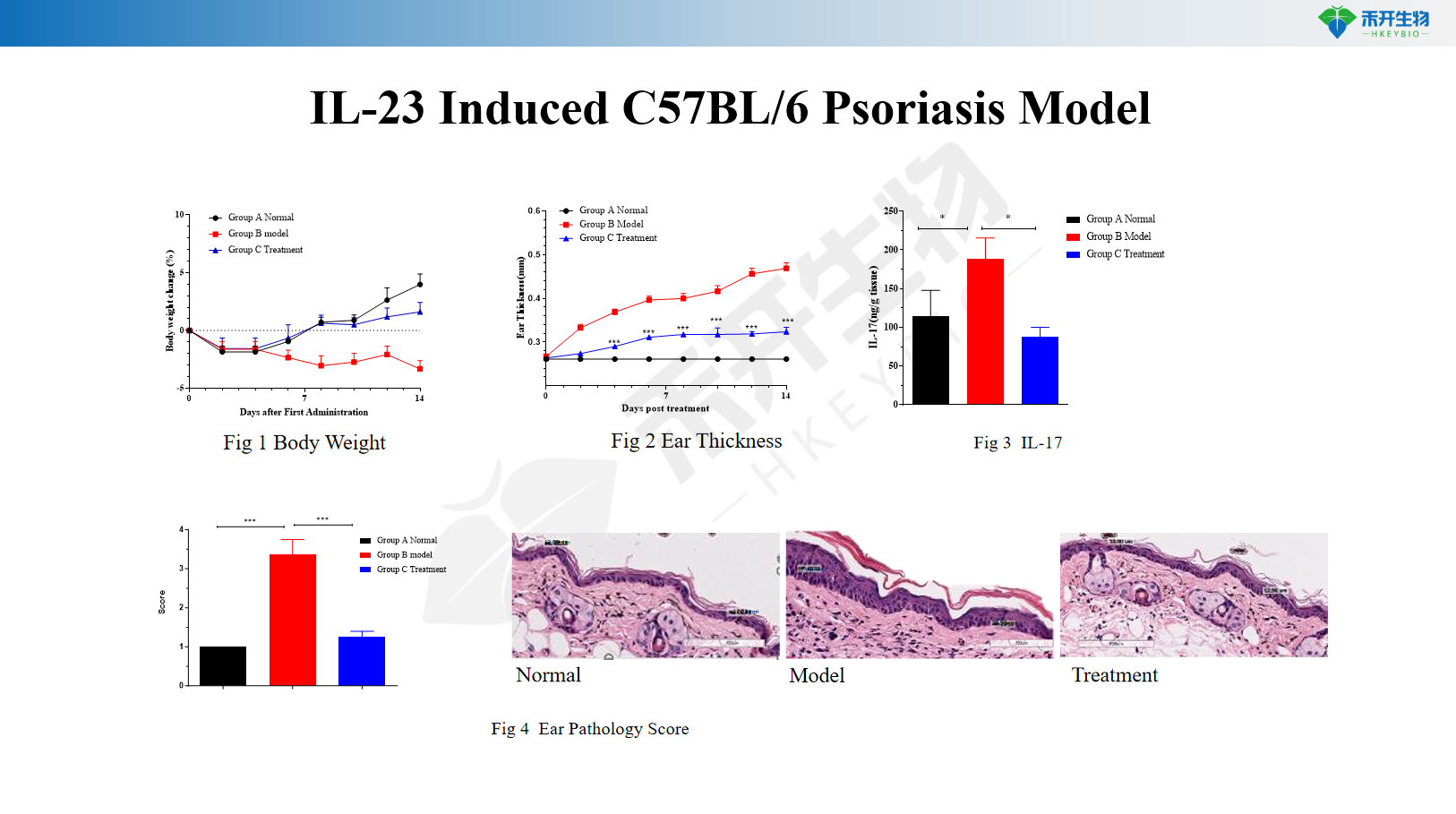
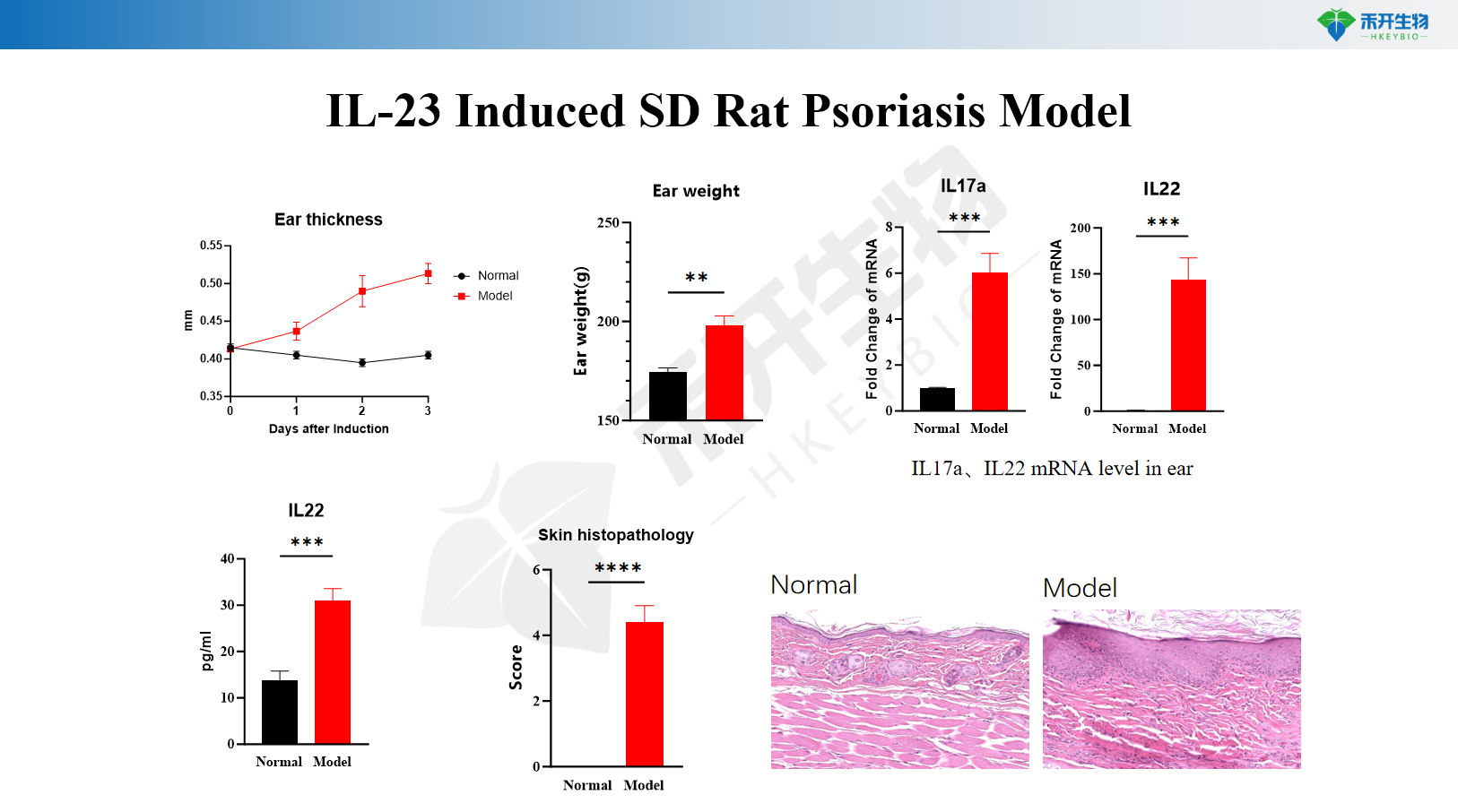
IL-23 Induced SD Rat Psoriasis Model
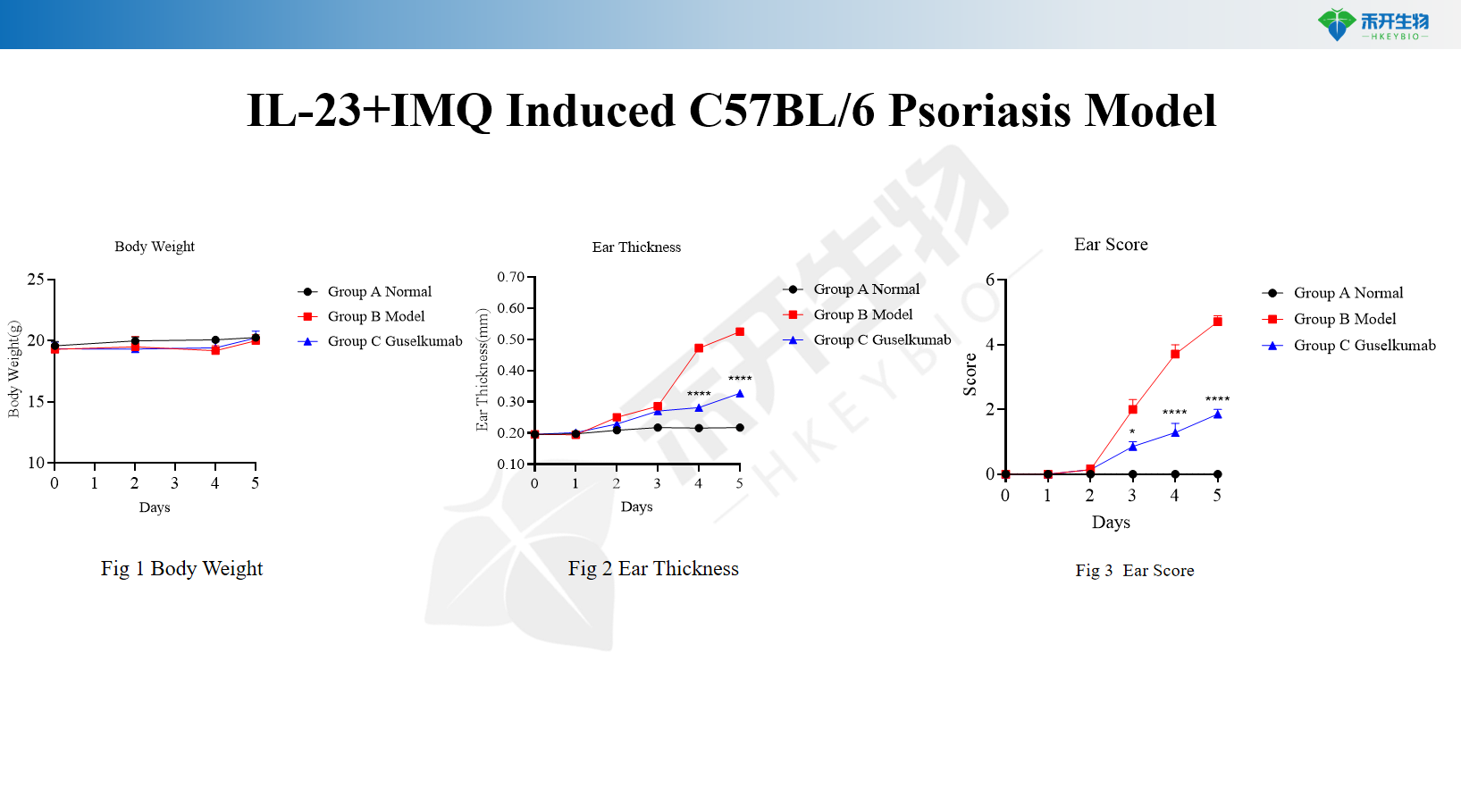
IL-23+IMQ Induced C57BL/6 Psoriasis Model
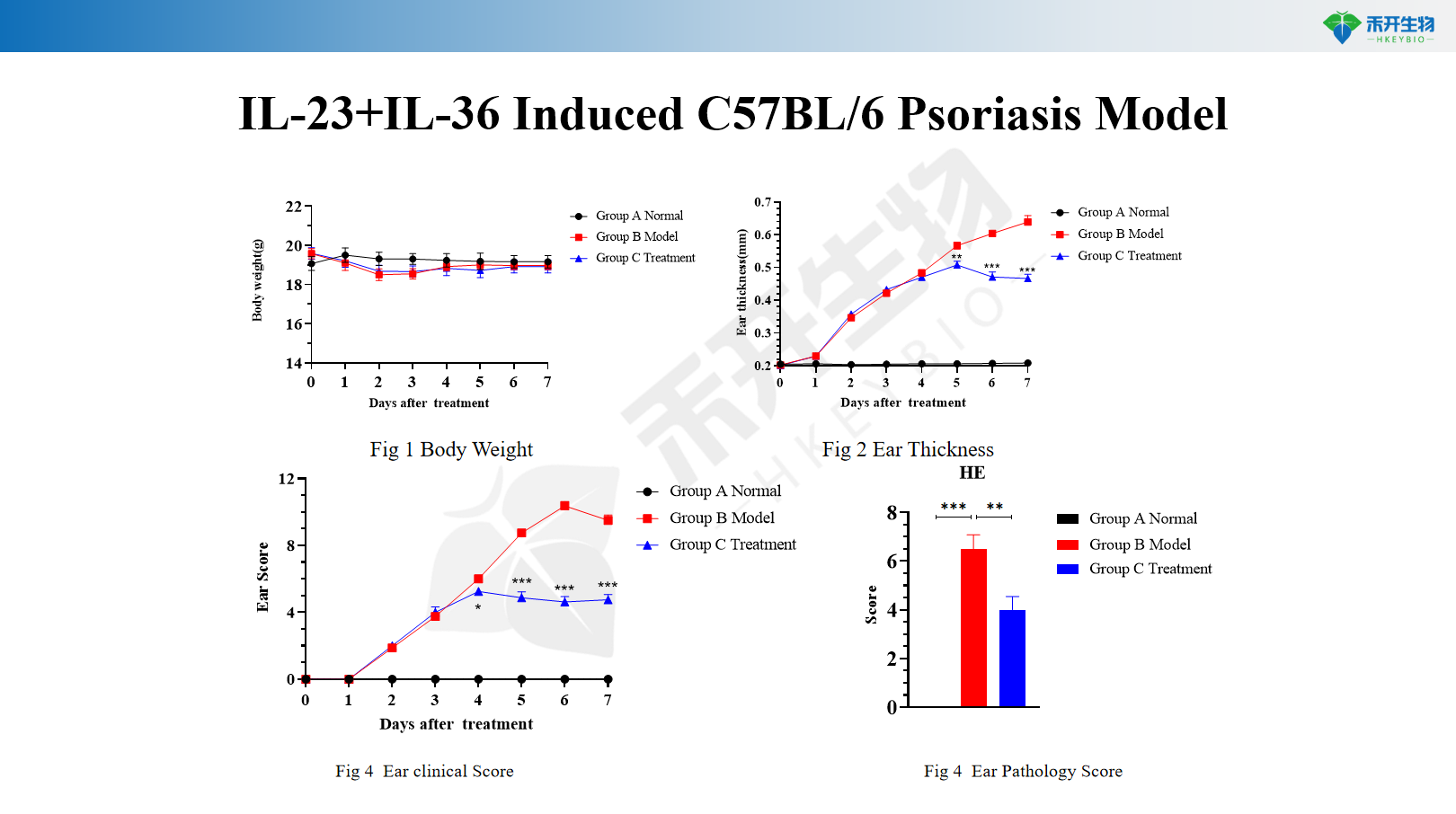
IL-23+IL-36 Induced C57BL/6 Psoriasis Model
Applications
Our psoriasis animal models serve a broad spectrum of drug discovery and development applications, from early target validation through IND-enabling toxicology. The versatility of these preclinical psoriasis models enables sponsors to evaluate diverse therapeutic modalities in a physiologically relevant context. Representative applications include:
• Efficacy testing of biologics targeting IL-17 (secukinumab, ixekizumab), IL-23 (guselkumab, risankizumab), and TNF-α (etanercept, adalimumab) in a physiologically relevant preclinical setting.
• Evaluation of small-molecule inhibitors including JAK inhibitors (tofacitinib, upadacitinib), PDE4 inhibitors (apremilast), TYK2 modulators, and emerging topical therapies entering dermatology pipelines.
• Target validation for Th17, IL-23, and TLR pathways in psoriasis, supporting go/no-go decision-making early in the drug discovery process and informing clinical biomarker strategy.
• Biomarker discovery and characterization (IL-17A quantification, PASI scoring algorithms, histopathology grading systems) to support patient stratification and clinical endpoint selection.
• IND-enabling pharmacology and toxicology studies conducted under GLP principles, providing regulatory agencies with comprehensive, audit-ready data packages for investigational new drug applications.
Model Specifications
Parameter | Specification |
Species/Strains | C57BL/6 mouse, BALB/c mouse, Wistar rat |
Induction methods | Topical IMQ cream (5%) daily for 5–7 days; Intradermal IL-23 injection (single or multiple doses); IMQ + IL-23 combination; IL-23 + IL-36 combination |
Study duration | 5–14 days (IMQ models); 3–7 days (IL-23 models) |
Key endpoints | PASI score (erythema, scaling, thickness), ear thickness, body weight, spleen index, IL-17A levels (ELISA/qPCR), skin histopathology (HE scoring for acanthosis, parakeratosis, inflammatory infiltration), optional: immunohistochemistry, flow cytometry |
| Positive control | Dexamethasone or anti-IL-17 antibody available as reference compounds |
| Data package | Raw data, analysis reports, clinical scores, histology slides, ELISA results, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What are the differences between IMQ and IL-23 induced psoriasis models?
A: IMQ activates TLR7/8, inducing IFN-α and downstream Th17 responses, mimicking the early phase of psoriasis. IL-23 directly activates γδ T cells and Th17 cells, more specifically targeting the IL-23/IL-17 axis central to chronic plaque psoriasis. Combination models provide enhanced severity and better recapitulate human disease.
Q: Which strain is best for psoriasis studies?
A: C57BL/6 mice are most commonly used for genetic studies and transgenic models. BALB/c mice exhibit stronger Th2 responses and are suitable for certain mechanistic studies. Wistar rats provide larger skin area for topical applications and histopathology.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different IMQ doses, IL-23 administration routes)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: IMQ models typically run 5–7 days; IL-23 models run 3–7 days; combination models may extend to 10–14 days.




























