Key Features & Benefits
Clinically relevant – Recapitulates human IgAN with mesangial IgA deposition, proteinuria, and glomerulonephritis.
Two strain options – BALB/c and C57BL/6 models available to suit different genetic backgrounds and experimental needs.
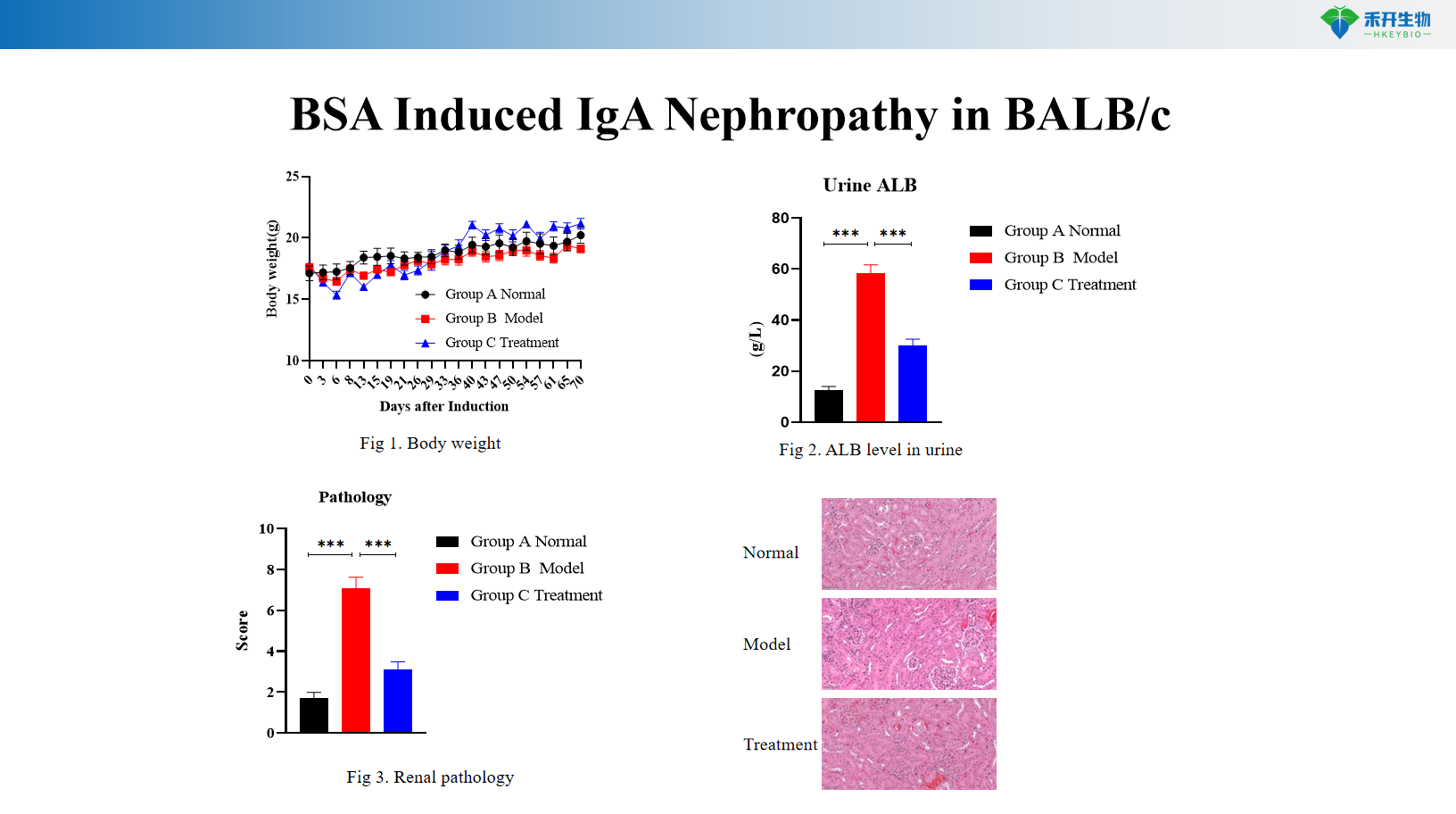
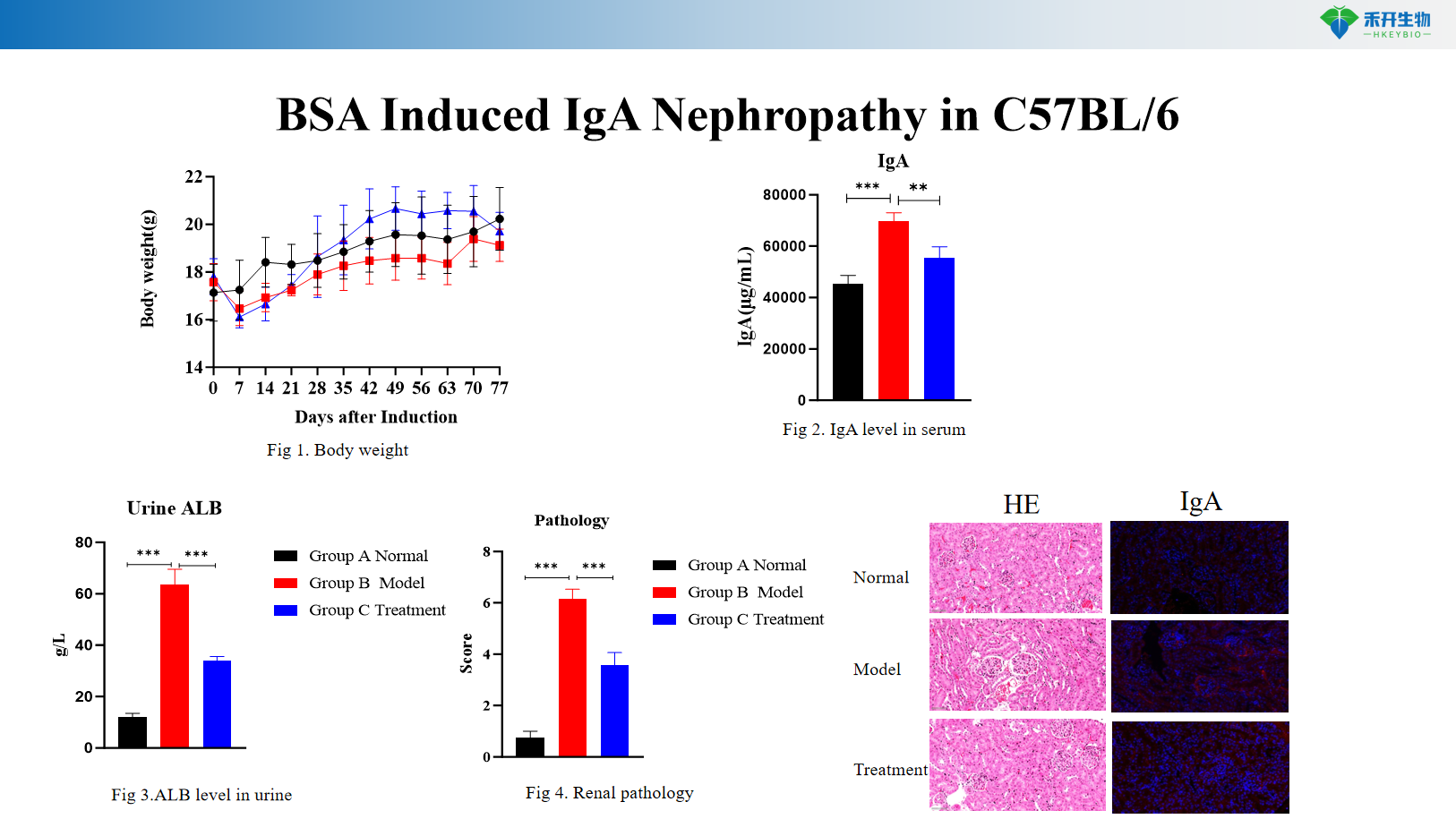
Comprehensive endpoints – Body weight, urinary albumin (ALB), serum IgA levels, renal histopathology (HE, IgA immunofluorescence).
Translational value – Ideal for testing immunomodulators, corticosteroids, and renoprotective agents for IgA nephropathy.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
BSA Induced IgA Nephropathy in BALB/c
BSA Induced IgA Nephropathy in C57BL/6
Applications
• Efficacy testing of immunomodulators (corticosteroids, mycophenolate, cyclophosphamide) for IgAN
• Evaluation of renoprotective agents and angiotensin-converting enzyme (ACE) inhibitors
• Target validation for IgA immune complex deposition and mesangial cell activation
• Biomarker discovery (albuminuria, serum IgA levels)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | BALB/c IgAN Model | C57BL/6 IgAN Model |
Species/Strain | BALB/c mouse | C57BL/6 mouse |
Induction method | Combined administration of bovine serum albumin (BSA) + carbon tetrachloride (CCl₄) + lipopolysaccharide (LPS) |
Study duration | 6–8 weeks | 6–8 weeks |
Key endpoints | Body weight, urinary albumin (ALB), renal histopathology (HE), mesangial IgA deposition | Body weight, urinary albumin (ALB), serum IgA levels, renal histopathology (HE), IgA immunofluorescence |
| Positive control | Corticosteroids (e.g., prednisolone) available as reference compounds |
Data package | Raw data, analysis reports, urine analysis, histology slides (HE, IgA IF), bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does BSA induce IgA nephropathy in mice?
A: BSA forms macromolecular aggregates that combine with IgA to form immune complexes. CCl₄ induces liver injury, promoting IgA production, while LPS acts as an adjuvant. These complexes deposit in the glomerular mesangium, triggering inflammation, proteinuria, and glomerulonephritis.
Q: What are the differences between BALB/c and C57BL/6 IgAN models?
A: Both strains develop mesangial IgA deposition and proteinuria. C57BL/6 mice may show more pronounced serum IgA elevation, making them suitable for studying systemic IgA responses. BALB/c mice are typically used for evaluating renal pathology and proteinuria endpoints.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different BSA doses, treatment timing)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Studies typically run 6–8 weeks post-induction, with proteinuria and histopathology assessed at endpoint.




























