Key Features & Benefits
Clinically relevant – UVB exposure in lupus-prone mice mimics photosensitive CLE, including skin lesions and systemic autoimmunity.
Comprehensive endpoints Translational value Technical Data & Validation
Technical Data & Validation
UVB Radiation Induced CLE Model in MRL/lpr Mice
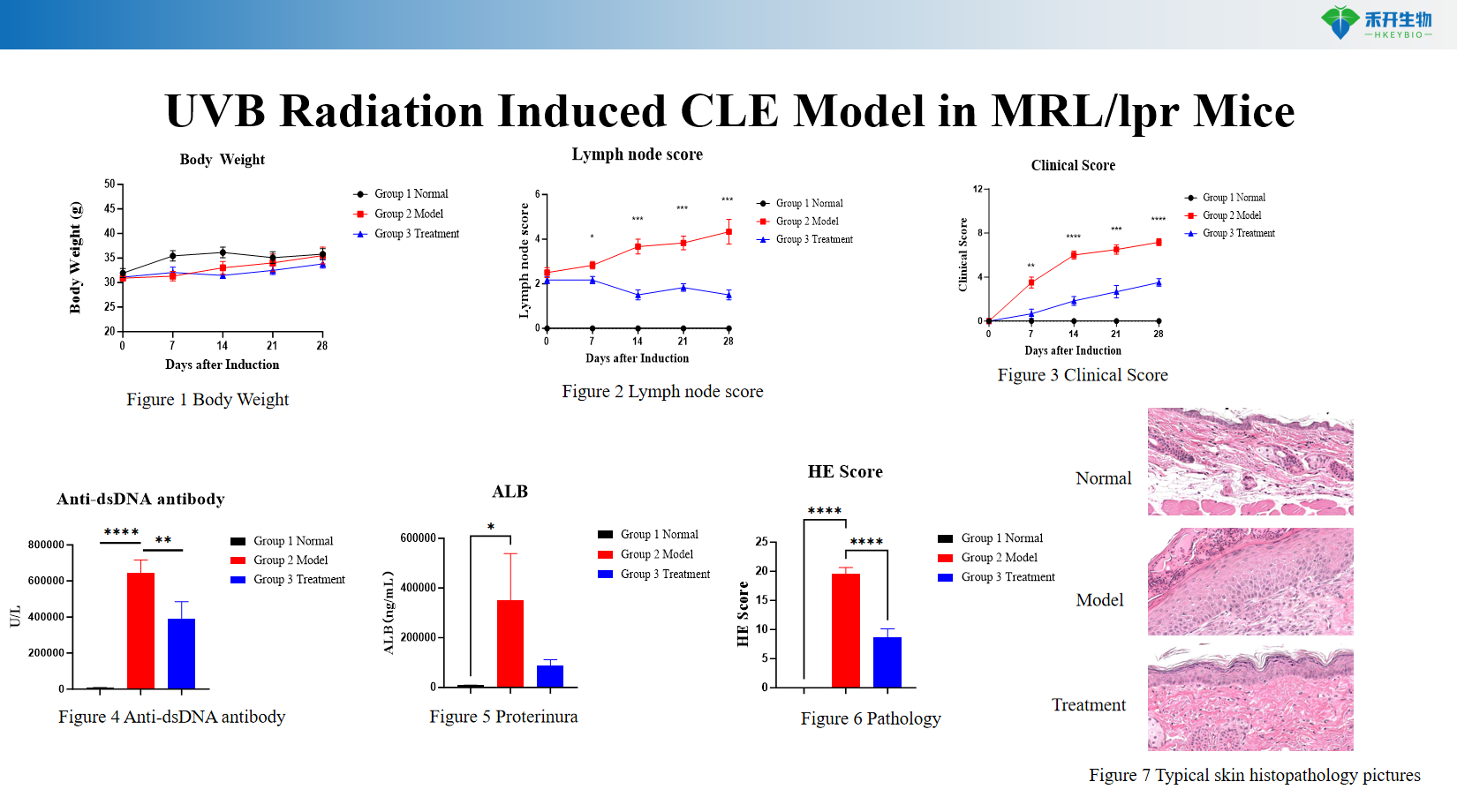
Applications
• Efficacy testing of topical and systemic immunomodulators (corticosteroids, calcineurin inhibitors, JAK inhibitors)
• Evaluation of antimalarials (hydroxychloroquine) and biologics targeting type I interferons (anifrolumab) or B cells (rituximab)
• Target validation for photosensitivity and autoimmune pathways in CLE
• Biomarker discovery (anti-dsDNA, interferon-stimulated genes)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | MRL/lpr mouse |
Induction method | Repeated UVB irradiation (312 nm, 100–200 mJ/cm²) on shaved dorsal skin, 3–5 times/week for 2–4 weeks |
Study duration | 3–6 weeks (irradiation + treatment phase) |
Key endpoints | Body weight, clinical skin score (erythema, scaling, excoriation), lymph node score, serum anti-dsDNA antibodies (ELISA), skin histopathology (HE with interface dermatitis scoring), optional: kidney histopathology, type I interferon signature, immunofluorescence for immune complex deposition |
Data package | Raw data, analysis reports, clinical scores, histology slides (skin, kidney), ELISA results, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: Why use MRL/lpr mice for the CLE model?
A: MRL/lpr mice spontaneously develop lupus-like autoimmunity with lymphadenopathy and anti-dsDNA antibodies. UVB exposure accelerates and localizes skin manifestations, making them ideal for studying photosensitive CLE.
Q: What are the key similarities with human CLE?
A: The model exhibits photosensitive skin lesions, interface dermatitis, elevated anti-dsDNA antibodies, and immune complex deposition, closely resembling human CLE (discoid, subacute, or acute forms).
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different UVB doses, treatment schedules)?
A: Absolutely. Our scientific team tailors UVB irradiation protocols, treatment schedules, and endpoint analyses to your specific drug candidate.















