Key Features & Benefits
Clinically relevant – Recapitulates human AD: skin inflammation, epidermal hyperplasia, Th2 bias, elevated IgE.
Three complementary models – DNCB, DNCB+IL-31, and HDM+SEB cover different AD endotypes.
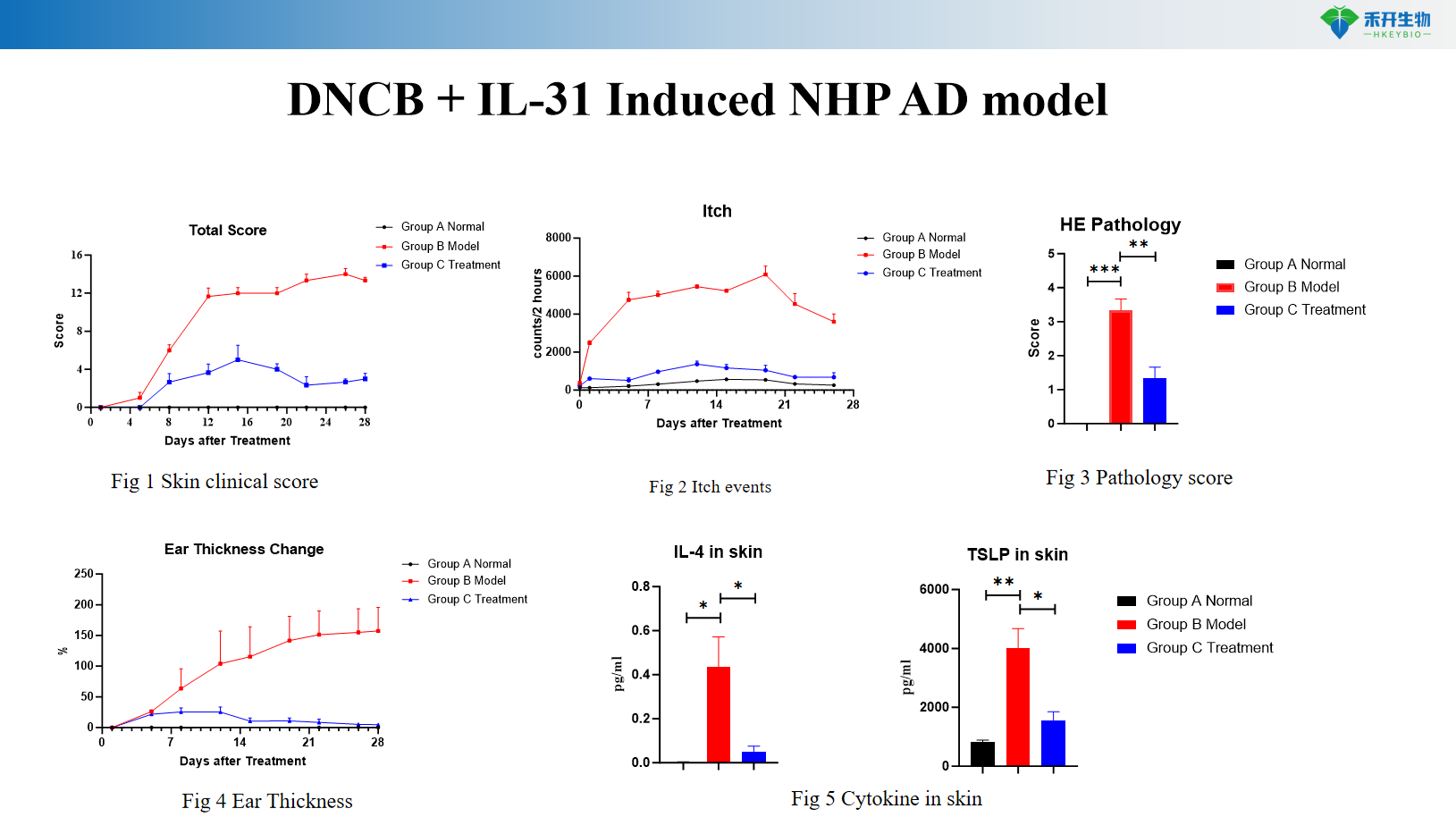
Pruritus quantification – Itch events scoring in IL-31 model to assess anti-pruritic efficacy.
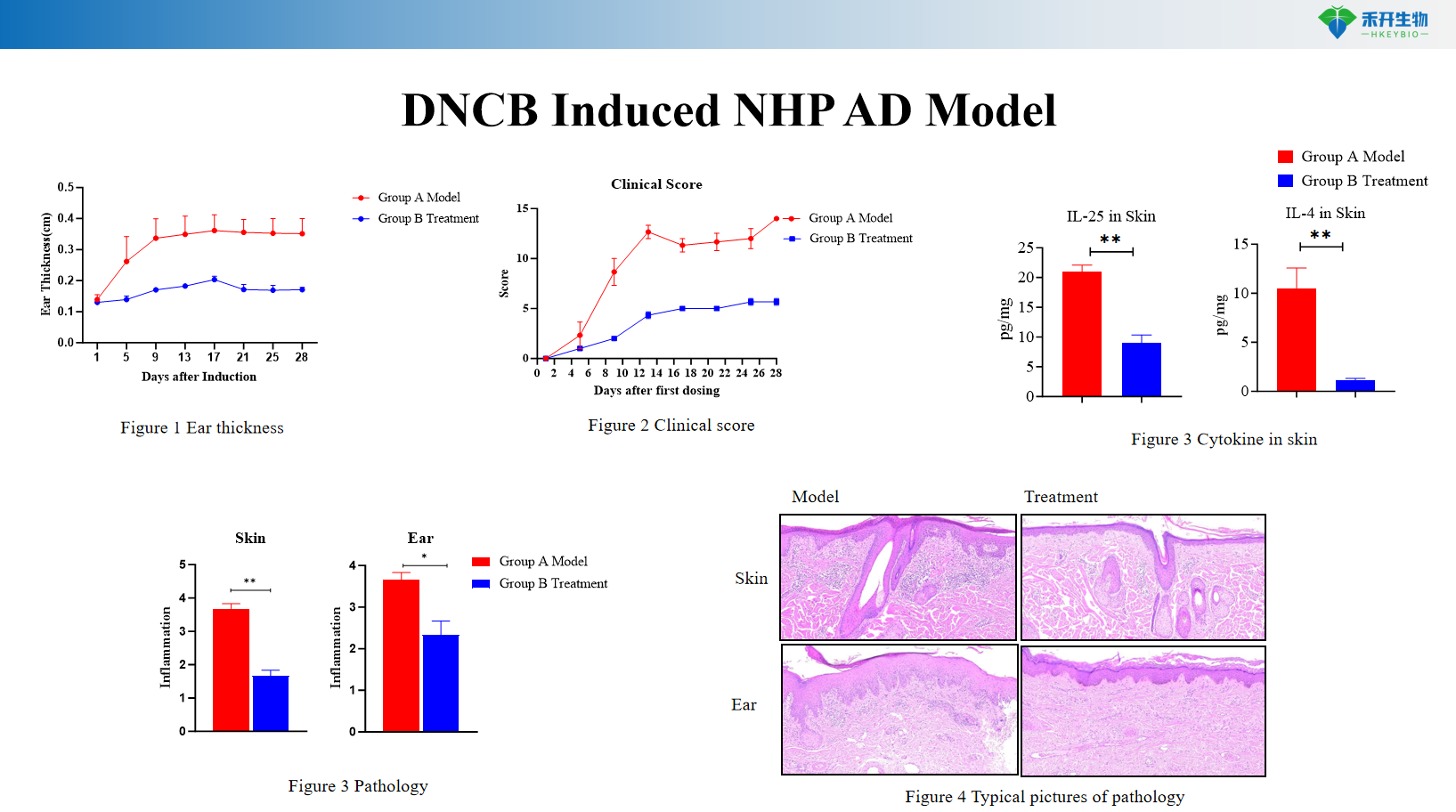
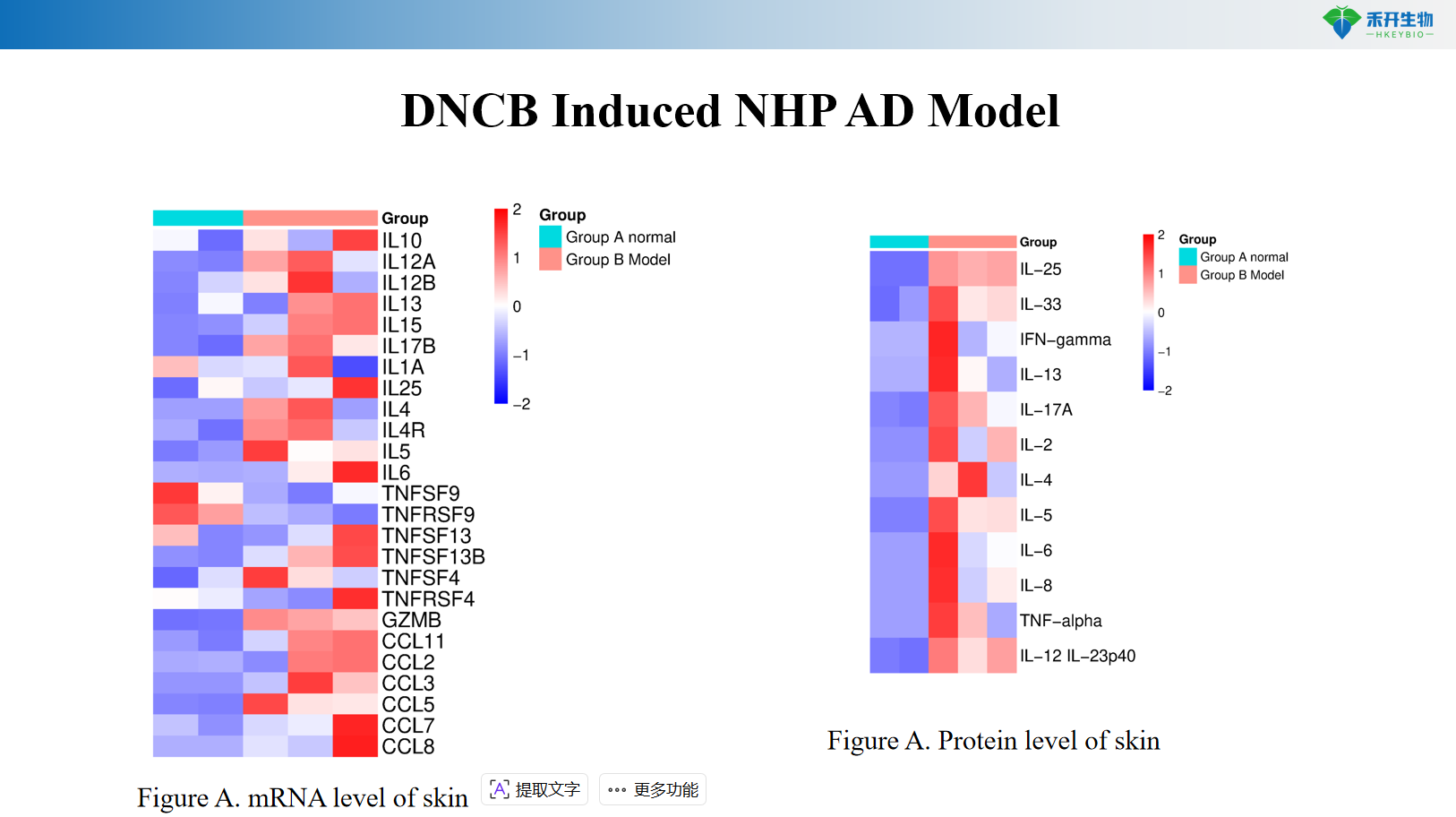
Multi-endpoint analysis – Clinical score, ear thickness, cytokine profiling (IL-4, IL-13, IL-31), histopathology, skin mRNA/protein.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Representative data from our NHP Atopic Dermatitis (AD) Model:
DNCB Induced NHP AD Model
DNCB + IL-31 Induced NHP AD model
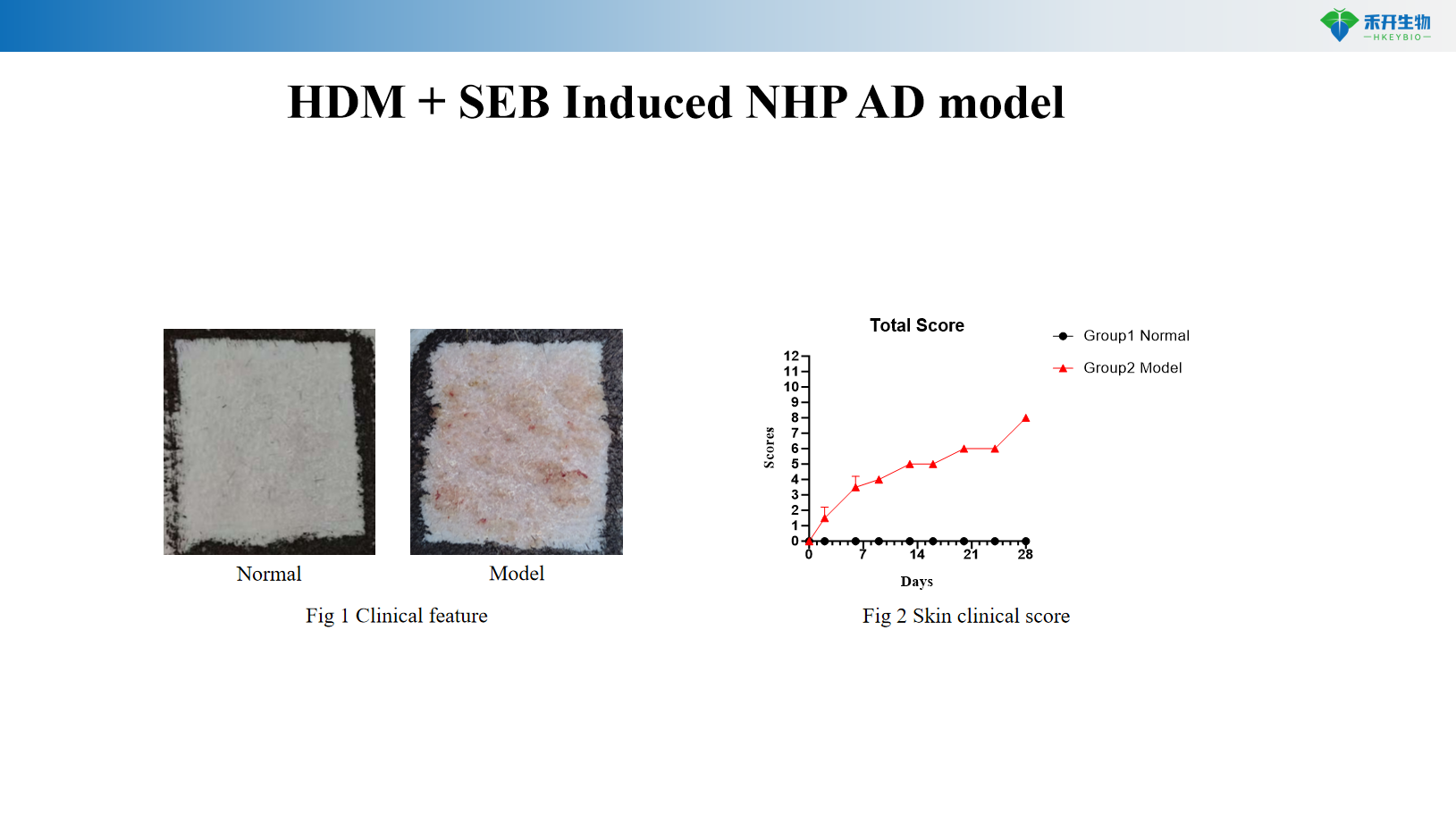
HDM + SEB Induced NHP AD model
Applications
•Efficacy testing of anti-inflammatory and anti-pruritic drugs (biologics, JAK inhibitors, topical agents)
• Target validation for Th2, Th17, Th22 pathways in AD
• Biomarker discovery (IgE, IL-4, IL-13, IL-31, TSLP)
• Mechanism of action (MOA) investigations
• IND-enabling toxicology and safety pharmacology studies
Model Specifications
Parameter | DNCB Induced Model | DNCB + IL-31 Induced Model | HDM+SEB Induced Model |
Species | Cynomolgus macaque | Cynomolgus macaque | Cynomolgus macaque |
Induction method | Repeated topical DNCB (2,4-dinitrochlorobenzene) | DNCB + IL-31 administration | HDM + SEB (Staphylococcal Enterotoxin B) |
Study duration | 4–6 weeks | 4–6 weeks (DNCB) + IL-31 dosing | 6–8 weeks |
Key endpoints | Clinical score, ear thickness, skin histopathology, Th2 cytokines (IL-4, IL-13), IgE, skin mRNA | Clinical score, itch events, ear thickness, skin cytokines (IL-4, IL-13), histopathology score | Clinical score, IgE, Th2/Th17/Th22 cytokines, skin histopathology |
Data package | Raw data, analysis reports, histology slides, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What are the differences between the three AD models?
A: DNCB model focuses on contact dermatitis-like inflammation; DNCB+IL-31 adds persistent pruritus to mimic the itch-scratch cycle; HDM+SEB model better represents allergic AD with IgE involvement and Th2/Th17/Th22 activation.
Q: Which model is best for testing anti-pruritic drugs?
A: The DNCB+IL-31 model is specifically designed to simulate chronic pruritus with quantifiable itch events, making it ideal for evaluating anti-itch efficacy.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols?
A: Absolutely. Our scientific team tailors dosing regimens, endpoint analyses, and model selection to your specific drug candidate and MOA.





















