Key Features & Benefits
Regulatory gold standard – TDAR is recommended by FDA, EMA, and ICH S8 as a first-choice immune function test for immunotoxicity assessment.
Comprehensive immune assessment Quantifiable endpoints – KLH-specific IgM and IgG titers; B cell phenotyping (percentage and absolute counts); primary and secondary response kinetics.
IND-ready data packages – Studies can be conducted in accordance with GLP principles for regulatory submissions.
Technical Data & Validation
Representative data from our KLH Induced NHP TDAR model:
KLH Induced NHP TDAR Model
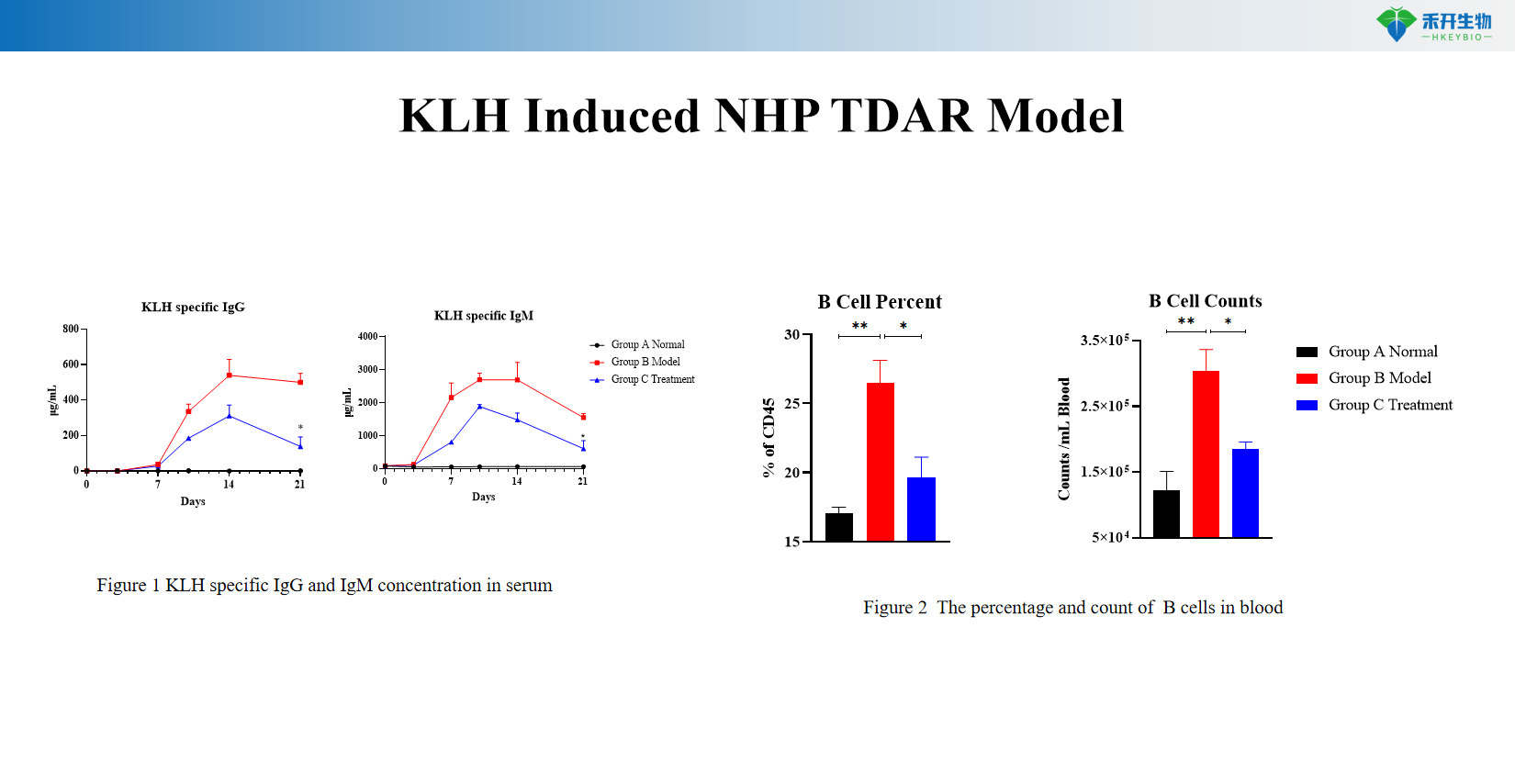
Applications
• Immunotoxicity assessment of small molecules, biologics, and immunomodulators
• IND-enabling studies requiring immune function evaluation (ICH S8 compliance)
• Mechanism of action studies for immunosuppressive or immunostimulatory drugs
• Vaccine adjuvant evaluation
• Biomarker discovery for immune competence
Model Specifications
Parameter | Specification |
Species | Cynomolgus macaque (Macaca fascicularis) |
Induction method | Immunization with keyhole limpet hemocyanin (KLH), typically with adjuvant, intramuscular or subcutaneous administration |
Study duration | 4–8 weeks (primary and secondary response assessment) |
Key endpoints | KLH-specific IgM (day 7-14), KLH-specific IgG (day 14-28), B cell phenotyping (percentage and absolute counts), optional: T cell subsets, cytokine profiling |
Data package | Raw data, analysis reports, flow cytometry files, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: Why is TDAR considered the gold standard for immunotoxicity assessment?
A: TDAR integrates multiple components of adaptive immunity (antigen presentation, T cell help, B cell activation, antibody production) into a single functional assay. Regulatory guidelines (ICH S8, FDA, EMA) recommend TDAR as a first-choice immune function test.
Q: Why use KLH as the antigen?
A: KLH is a highly immunogenic protein with no prior exposure in humans or laboratory animals, ensuring that measured antibody responses are specific to the immunization and not confounded by pre-existing immunity.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles to support regulatory submissions (FDA, EMA) and meet ICH S8 immunotoxicity testing requirements.
Q: Do you offer customized study protocols (e.g., different adjuvants, timing of immunizations)?
A: Absolutely. Our scientific team tailors immunization schedules, adjuvant selection, and endpoint analyses to your specific drug candidate and regulatory requirements.


















