Main features and benefits
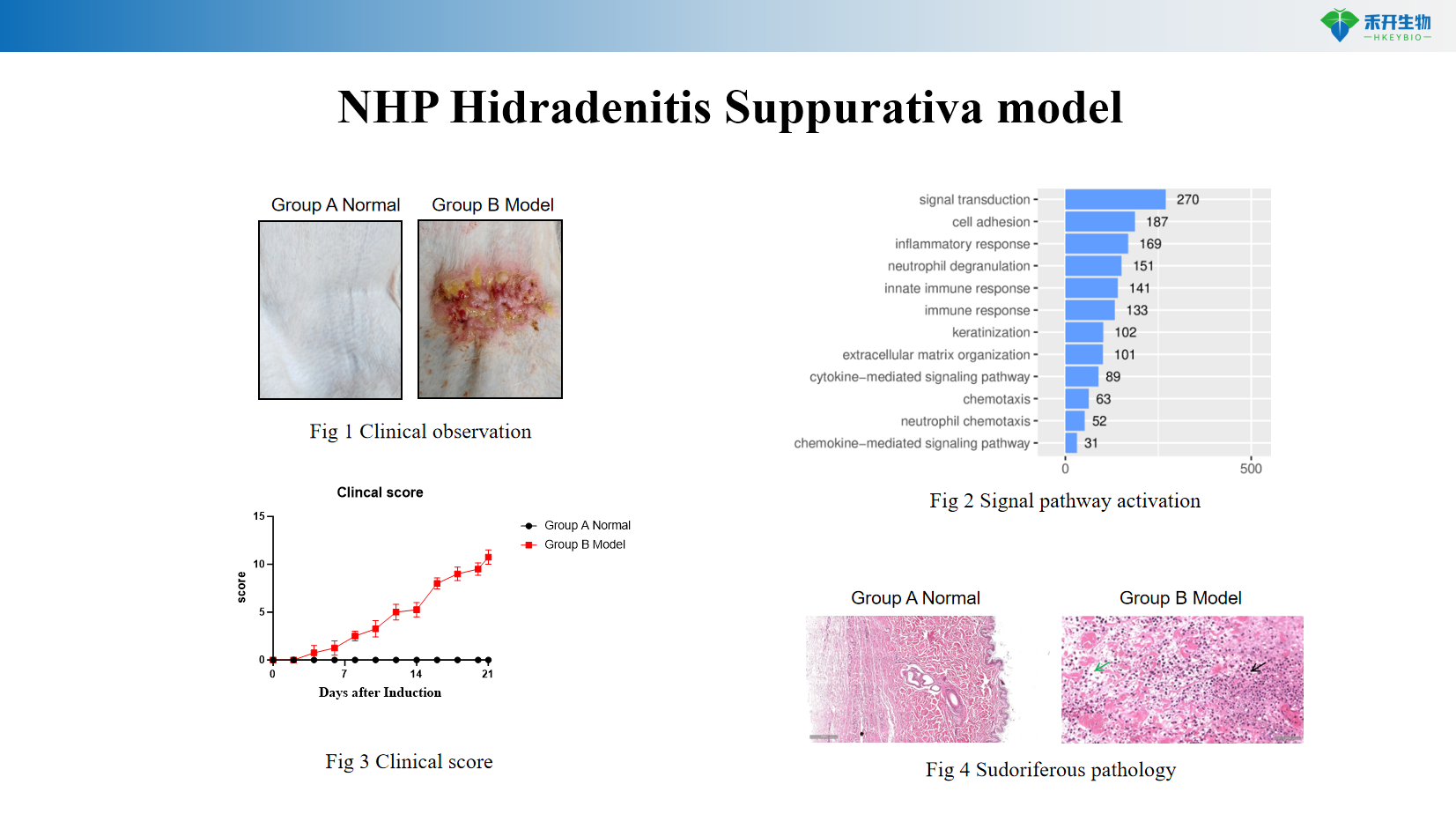
Clinically relevant – Mimics human HS: dermal tunnels, neutrophil infiltration, chronic inflammatory skin lesions.
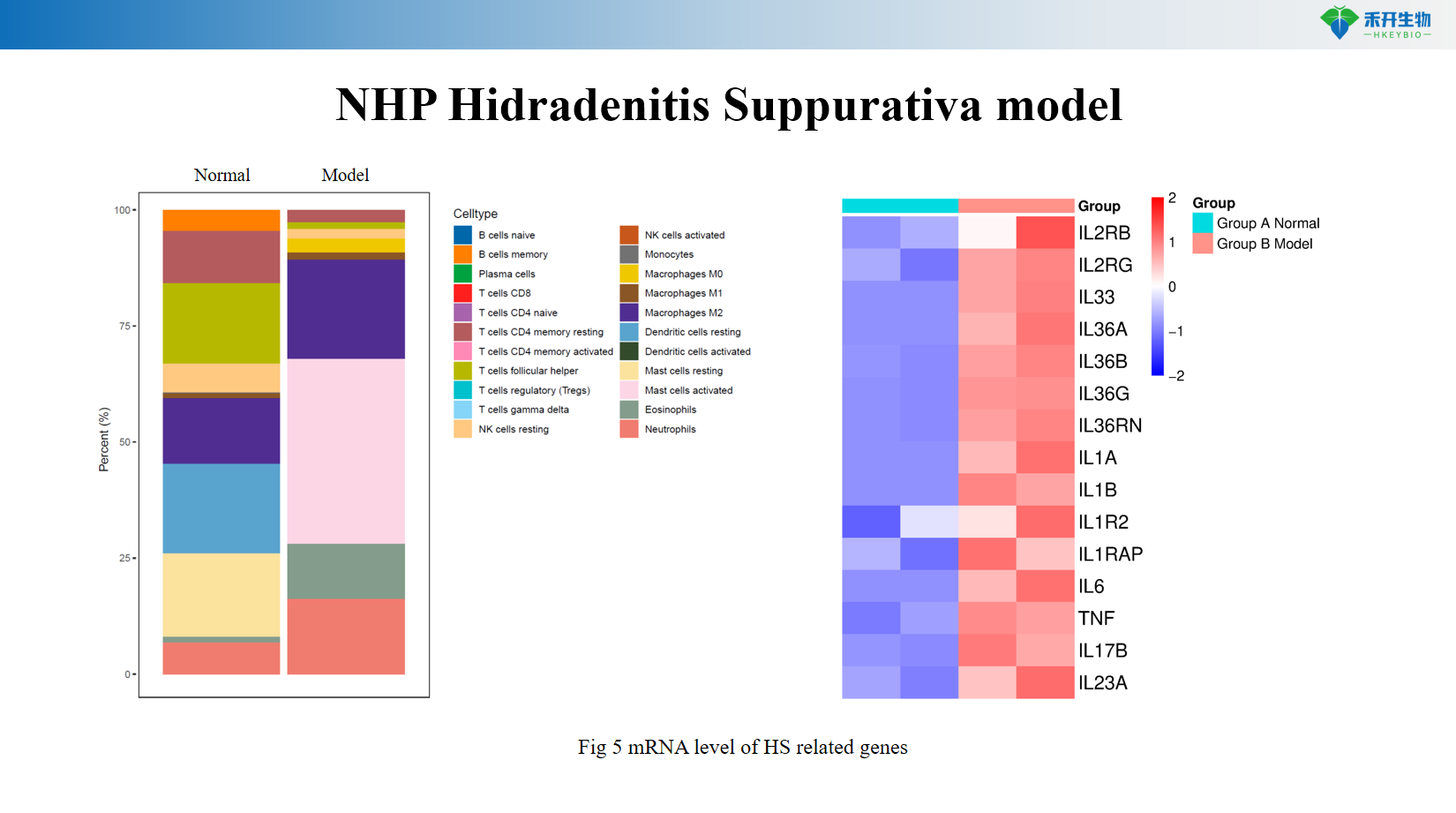
Multi-mechanism involved – recapitulates innate (IL-1β, TNF-α, neutrophils) and adaptive (Th1/Th17, B cells) immune components.
Comprehensive endpoint - clinical observation, histopathology, and mRNA expression of HS-related genes (IL-1β, TNF-α, CXCL1, CXCL8, Th17 markers).
Translational Value – Ideal for testing biologics (anti-TNF, anti-IL-1, anti-IL-17), JAK inhibitors, and small molecules.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Representative data for our NHP HS model:
NHP hidradenitis suppurativa model
Application areas
• Efficacy testing of biologics (anti-TNF, anti-IL-1, anti-IL-17/23), JAK inhibitors, and small molecule anti-inflammatory drugs
• Target validation of innate pathways and Th1/Th17 pathways in HS
• Biomarker discovery (gene signatures, soluble mediators)
• Mechanism of action (MOA) studies
• Toxicology and safety pharmacology studies to support IND
Model specifications
scope | Specification |
Species | cynomolgus monkey ( Macaca fascicularis ) |
induction method | Proprietary multi-step protocol (combination of mechanical/chemical/immunostimulation) – details available in CDA |
study time | 6–10 weeks (induction + treatment phase) |
critical endpoint | Clinical score (erythema, nodules, tunnels), histopathology (dermal tunnels, inflammatory infiltrates), mRNA expression of IL-1β, TNF-α, CXCL1, CXCL8, Th17 markers, immunohistochemistry |
packet | Raw data, analysis reports, histological sections, gene expression data, bioinformatics (optional) |
❓ FAQ
Q: What is unique about this NHP HS model?
A: It outlines the complex immune landscape of human HS, including innate (neutrophils, macrophages) and adaptive (Th1/Th17, B cells) components, as well as the formation of dermal tunnels and upregulation of key inflammatory mediators.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized research plans?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.
Q: What are the typical readings used to evaluate treatment effectiveness?
A: Clinical scoring of skin lesions, histopathological assessment of dermal channels and inflammatory infiltrates, and qPCR of HS-related gene expression (IL-1β, TNF-α, CXCL1, CXCL8, Th17 markers).




















