Key Features & Benefits
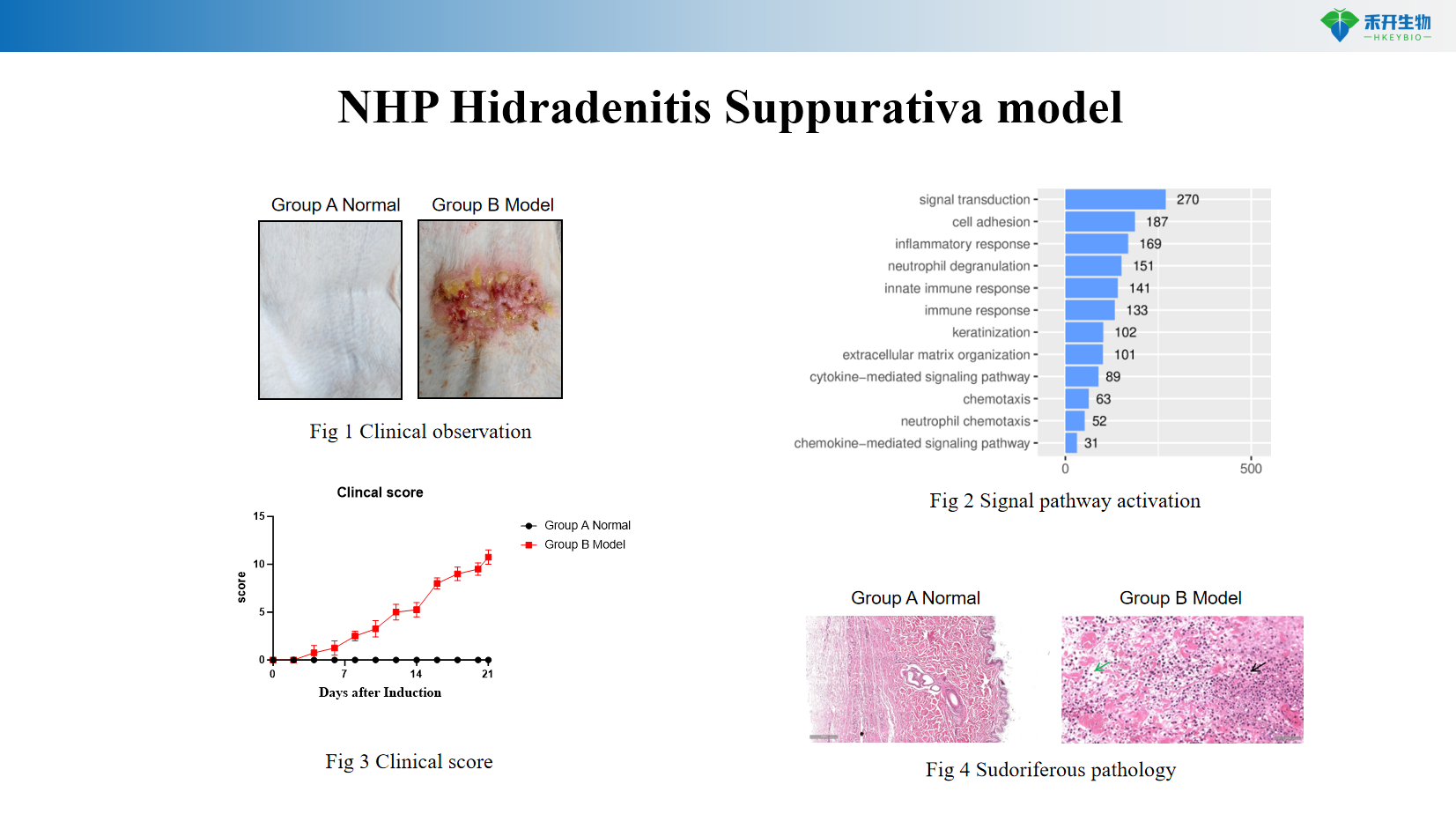
Clinically relevant – Mimics human HS: dermal tunnels, neutrophilic infiltration, chronic inflammatory skin lesions.
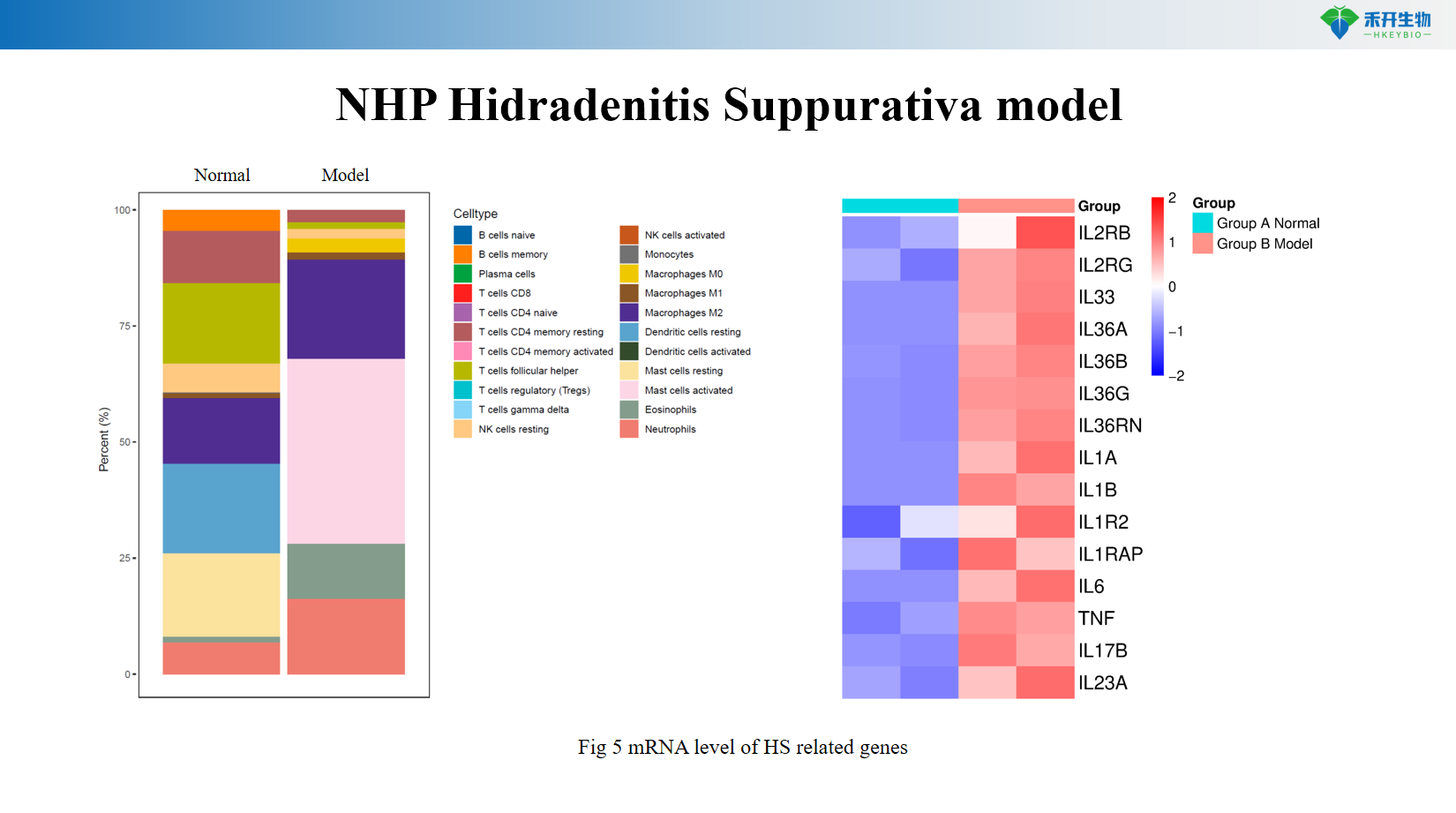
Multi-mechanism engagement – Recapitulates innate (IL-1β, TNF-α, neutrophils) and adaptive (Th1/Th17, B-cell) immune components.
Comprehensive endpoints – Clinical observation, histopathology, mRNA expression of HS-related genes (IL-1β, TNF-α, CXCL1, CXCL8, Th17 markers).
Translational value – Ideal for testing biologics (anti-TNF, anti-IL-1, anti-IL-17), JAK inhibitors, and small molecules.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Representative data from our NHP HS model:
NHP Hidradenitis Suppurativa model
Applications
• Efficacy testing of biologics (anti-TNF, anti-IL-1, anti-IL-17/23), JAK inhibitors, and small molecule anti-inflammatory drugs
• Target validation for innate and Th1/Th17 pathways in HS
• Biomarker discovery (gene signatures, soluble mediators)
• Mechanism of action (MOA) studies
• IND-enabling toxicology and safety pharmacology studies
Model Specifications
Parameter | Specification |
Species | Cynomolgus macaque (Macaca fascicularis) |
Induction method | Proprietary multi-step protocol (combination of mechanical/chemical/immunological stimuli) – details available under CDA |
Study duration | 6–10 weeks (induction + treatment phase) |
Key endpoints | Clinical score (erythema, nodules, tunnels), histopathology (dermal tunnels, inflammatory infiltrates), mRNA expression of IL-1β, TNF-α, CXCL1, CXCL8, Th17 markers, immunohistochemistry |
Data package | Raw data, analysis reports, histology slides, gene expression data, bioinformatics (optional) |
❓ Frequently Asked Questions
Q:What makes this NHP HS model unique?
A: It recapitulates the complex immune landscape of human HS, including both innate (neutrophils, macrophages) and adaptive (Th1/Th17, B-cell) components, with formation of dermal tunnels and upregulation of key inflammatory mediators.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What are the typical readouts for assessing therapeutic efficacy?
A: Clinical scoring of skin lesions, histopathological evaluation of dermal tunnels and inflammatory infiltrates, and qPCR for HS-related gene expression (IL-1β, TNF-α, CXCL1, CXCL8, Th17 markers).




















