Key Features & Benefits
Clinically relevant – Recapitulates human hyperuricemia with elevated serum uric acid, suitable for studying gout and uric acid nephropathy.
Mechanism-driven – Potassium oxonate inhibits uricase (the enzyme that degrades uric acid in rodents), mimicking human uric acid metabolism; adenine increases purine load.
Quantifiable endpoint – Serum uric acid levels measured by colorimetric or enzymatic assays.
Translational value – Ideal for testing xanthine oxidase inhibitors (allopurinol, febuxostat), uricosuric agents, and novel urate-lowering compounds.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
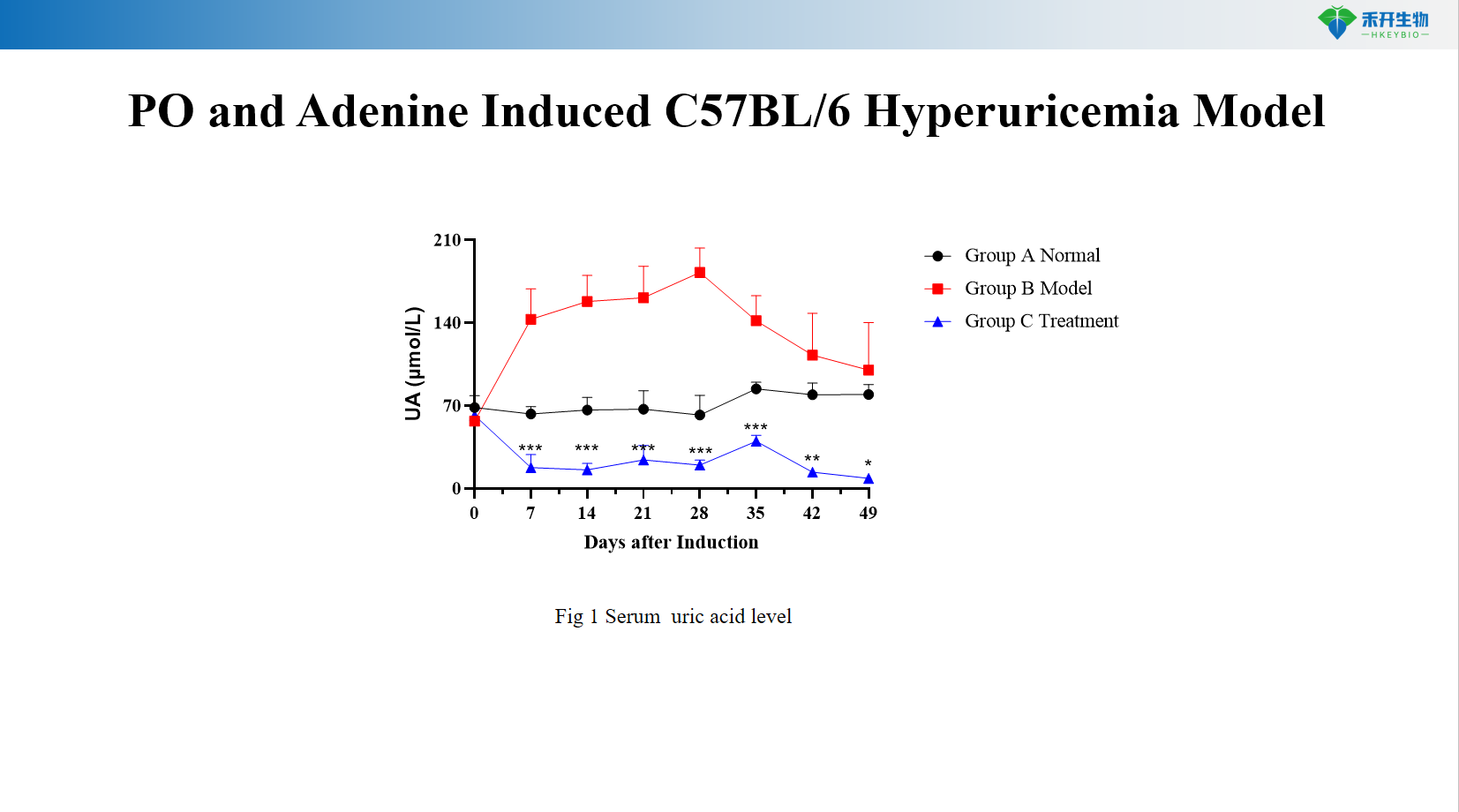
PO and Adenine Induced C57BL/6 Hyperuricemia Model
Applications
• Efficacy testing of xanthine oxidase inhibitors (allopurinol, febuxostat, topiroxostat)
• Evaluation of uricosuric agents (probenecid, benzbromarone, lesinurad)
• Target validation for uric acid metabolism pathways
• Biomarker discovery (uric acid, xanthine oxidase activity, renal function markers)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | C57BL/6 mouse |
Induction method | Oral administration of potassium oxonate (250–500 mg/kg) + adenine (50–100 mg/kg), daily for 7–14 days, often combined with high-purine diet |
Study duration | 1–3 weeks (induction + treatment phase) |
Key endpoints | Serum uric acid levels, optional: serum creatinine, BUN, xanthine oxidase activity, renal histopathology, urinary uric acid excretion |
| Positive control | Allopurinol (xanthine oxidase inhibitor) available as reference compound |
Data package | Raw data, analysis reports, clinical chemistry results, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: Why use potassium oxonate and adenine to induce hyperuricemia?
A: Rodents have uricase, an enzyme that degrades uric acid to allantoin, resulting in very low baseline uric acid levels. Potassium oxonate inhibits uricase, elevating uric acid to human-like levels. Adenine increases purine load and promotes uric acid production, enhancing the hyperuricemic effect.
Q: What are the key similarities with human hyperuricemia?
A: The model exhibits significantly elevated serum uric acid levels and responds to standard xanthine oxidase inhibitors like allopurinol, closely mimicking human hyperuricemia and providing a predictive platform for urate-lowering therapies.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different PO/adenine doses, combination with high-purine diet)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Pilot studies are typically completed within 14 days, including induction, treatment, and serum uric acid measurement at endpoint.















