Main features and benefits
Etiologically related – caused by cigarette smoke and the leading cause of chronic obstructive pulmonary disease in humans.
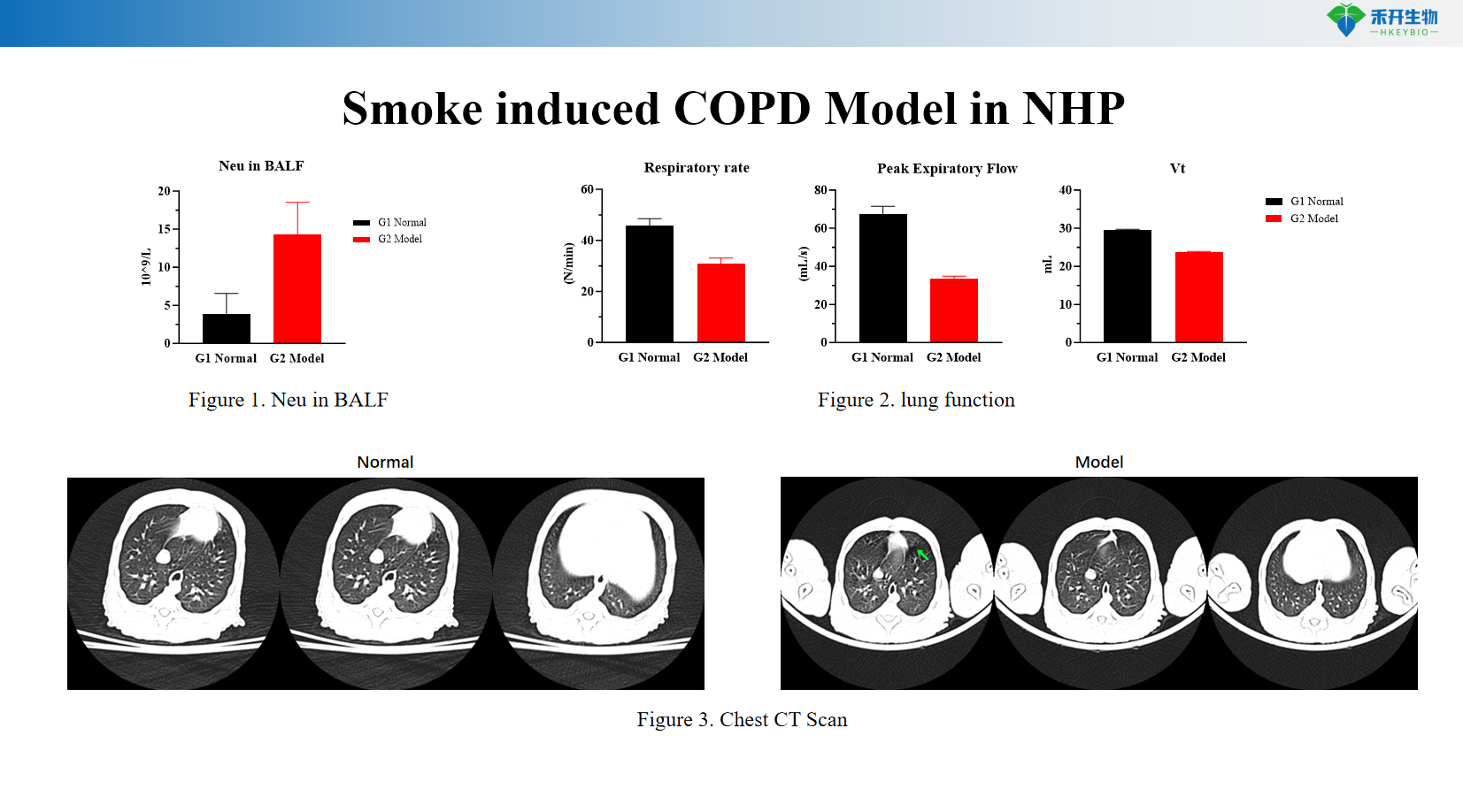
Clinically translatable endpoints – Pulmonary function testing, chest CT imaging, BALF cytology (neutrophils), weight monitoring.
Chronic inflammation —reproducible airway neutrophilia and structural lung damage.
Multisystem evaluation – evaluates the pulmonary and systemic effects of test compounds.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Representative data from our cigarette smoke-induced NHP COPD model:
Smoke-induced COPD model in NHP
Application areas
• Efficacy testing of anti-inflammatory drugs for COPD (e.g. PDE4 inhibitors, corticosteroids, biologics)
• Target validation in neutrophil-mediated airway disease
• Biomarker discovery (BALF cytology, cytokines, imaging biomarkers)
• Mechanism of action (MOA) studies
• Toxicology and safety pharmacology studies to support IND
Model specifications
scope | Specification |
Species | cynomolgus monkey ( Macaca fascicularis ) |
induction method | Chronic total body or nose-only exposure to cigarette smoke (daily, 3-6 months) |
study time | 3-6 months (induction period) + treatment period |
critical endpoint | Weight, lung function (spirometry), BALF cytology (neutrophils), chest CT imaging, histopathology (optional) |
packet | Raw data, analysis reports, CT images, BALF cytology reports, bioinformatics (optional) |
❓ FAQ
Q: How is the COPD model induced in NHP?
A: Mimicking the major human cause of COPD through months of chronic exposure to cigarette smoke.
Q: What are the main pathological characteristics of this model?
A: Airway inflammation (neutrophilia in BALF), progressive lung function decline, and emphysematous changes visible on CT imaging.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: How long does a typical efficacy study take?
A: The total study duration, including the COPD induction and treatment phases, is approximately 4-8 months, depending on the endpoint and study design.
















