Key Features & Benefits
Clinically relevant – Spontaneous autoimmune model closely mimics human autoimmune neuropathy with progressive sensory deficits and pain.
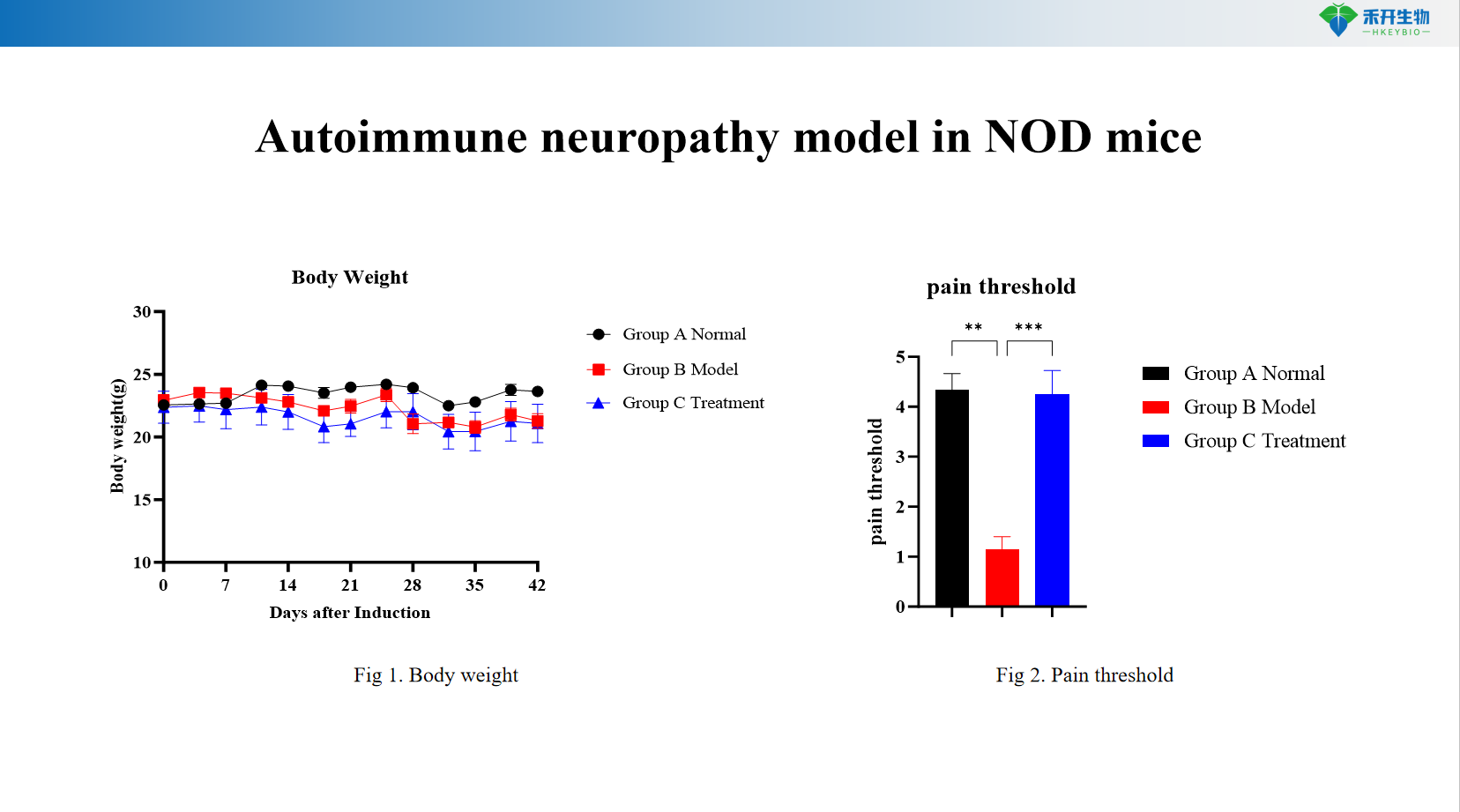
Quantifiable endpoints – Body weight monitoring, pain threshold measurement (von Frey filaments), sensory function tests.
Mechanism-driven – Autoimmune attack on peripheral nerves mediated by T cells and autoantibodies, mirroring human disease pathogenesis.
Translational value – Ideal for testing immunomodulators (corticosteroids, IVIG, rituximab), pain relievers (gabapentin, pregabalin), and neuroprotective agents.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Autoimmune neuropathy model in NOD mice
Applications
• Efficacy testing of immunomodulators (corticosteroids, IVIG, rituximab, mycophenolate) for autoimmune neuropathy
• Evaluation of neuropathic pain treatments (gabapentin, pregabalin, tricyclic antidepressants, SNRIs)
• Target validation for autoimmune-mediated nerve damage and pain pathways
• Biomarker discovery (autoantibodies, cytokine profiles, pain-related mediators)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | NOD mouse |
Induction method | Spontaneous autoimmune development (genetic susceptibility); onset typically occurs with age |
Study duration | 8–20 weeks (monitoring from pre-onset to established disease) |
Key endpoints | Body weight, pain threshold (von Frey mechanical allodynia), sensory function tests (hot plate, cold sensitivity), optional: nerve conduction velocity, histopathology of peripheral nerves (axonal degeneration, demyelination), immunohistochemistry (CD3, CD68, Iba1), serum autoantibodies (anti-neuronal antibodies) |
Data package | Raw data, analysis reports, behavioral data, histology slides, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does the NOD mouse model develop autoimmune neuropathy?
A: NOD mice have a genetic predisposition to autoimmune disorders. They spontaneously develop immune responses against peripheral nerve components, leading to T cell and autoantibody-mediated damage to axons and myelin, resulting in progressive sensory deficits and pain.
Q: What are the key similarities with human autoimmune neuropathy?
A: The model exhibits progressive weight loss, mechanical allodynia, sensory dysfunction, and histopathological evidence of nerve inflammation and damage, closely resembling human autoimmune peripheral neuropathy.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different ages at start, combination with pain relievers)?
A: Absolutely. Our scientific team tailors monitoring schedules, treatment protocols, and endpoint analyses to your specific drug candidate.































