Key Features & Benefits
Clinically relevant – Recapitulates human autoimmune uveitis with Th1/Th17 mediated inflammation, retinal damage, and blood-retinal barrier breakdown.
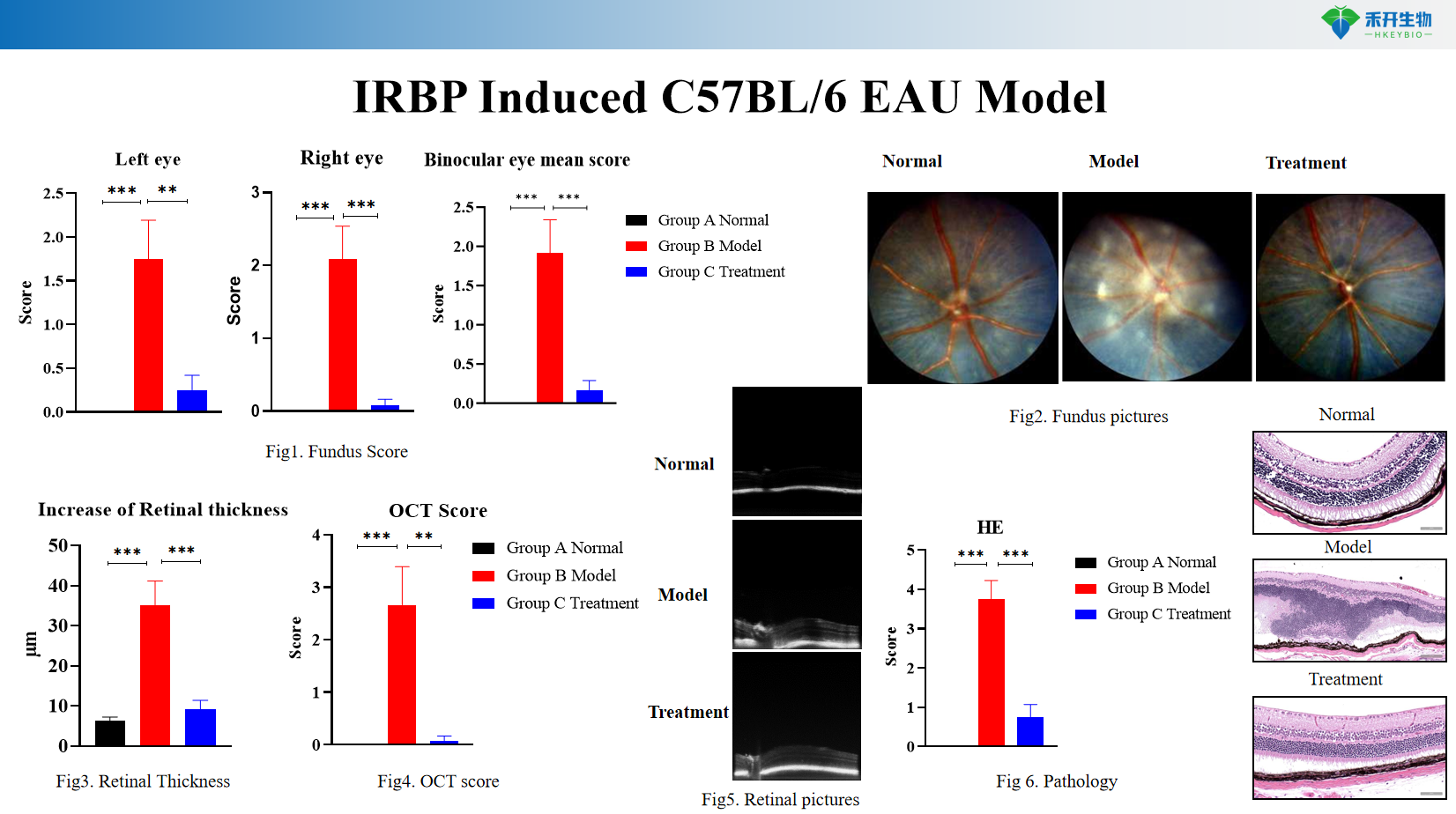
Advanced imaging endpoints – Fundus photography and scoring, optical coherence tomography (OCT) for retinal thickness measurement.
Comprehensive pathology – Histopathology (HE staining) for inflammatory infiltration and retinal structural damage.
Mechanism-driven – IRBP activates antigen-presenting cells, driving Th1 and Th17 differentiation and inflammatory cytokine production (IFN-γ, IL-17, TNF-α).
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
IRBP Induced C57BL/6 EAU Model
Applications
• Efficacy testing of immunomodulators (corticosteroids, methotrexate, mycophenolate) for autoimmune uveitis
• Evaluation of biologics targeting Th1/Th17 pathways (anti-IL-17, anti-IL-23, anti-IFN-γ)
• Target validation for T cell mediated autoimmune responses in the eye
• Biomarker discovery (inflammatory cytokines, retinal damage markers)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | C57BL/6 mouse |
Induction method | Subcutaneous immunization with IRBP peptide (e.g., IRBP1-20, 200-300 μg) emulsified in CFA supplemented with Mycobacterium tuberculosis, plus intraperitoneal pertussis toxin (0.5-1 μg) at immunization |
Study duration | 21–35 days post-immunization (peak disease ~14-21 days) |
Key endpoints | Fundus photography and clinical scoring (0-4 scale for optic disc, vasculitis, infiltrates), optical coherence tomography (OCT) for retinal thickness measurement, OCT scoring, histopathology (HE staining of retina for inflammatory infiltration and structural damage), optional: flow cytometry of retinal/lymph node immune cells (CD4+ T cells, Th1/Th17 subsets), cytokine analysis (IFN-γ, IL-17, TNF-α) |
Data package | Raw data, analysis reports, fundus images, OCT data, histology slides, bioinformatics (optional)Raw data, analysis reports, clinical scores, histology slides, serum analysis (IL-6, CRP), optional: anti-CII antibodies, micro-CT imaging |
❓ Frequently Asked Questions
Q: How does IRBP induce EAU?
A: IRBP is a retinal antigen. Immunization with IRBP peptide in adjuvant activates antigen-presenting cells, which present the antigen to naive T cells, driving their differentiation into pathogenic Th1 and Th17 effectors. These cells infiltrate the eye, release inflammatory cytokines (IFN-γ, IL-17, TNF-α), and cause retinal inflammation and damage.
Q: What are the key similarities with human autoimmune uveitis?
A: The model exhibits T cell-mediated inflammation (Th1/Th17), breakdown of blood-retinal barrier, inflammatory cell infiltration, retinal structural damage, and clinical signs visible by fundoscopy and OCT, closely mirroring human non-infectious uveitis.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different IRBP peptides, doses, timing of treatment)?
A: Absolutely. Our scientific team tailors immunization protocols, treatment schedules, and endpoint analyses to your specific drug candidate.



















