Main features and benefits
Clinically Relevant – Overview of human PN and IgE-mediated inflammation, chronic pruritus, and papulonodular skin lesions.
Mechanism driven - IgE and Th2 cytokine (IL-4)-mediated inflammation drives ongoing scratching and fibrotic changes.
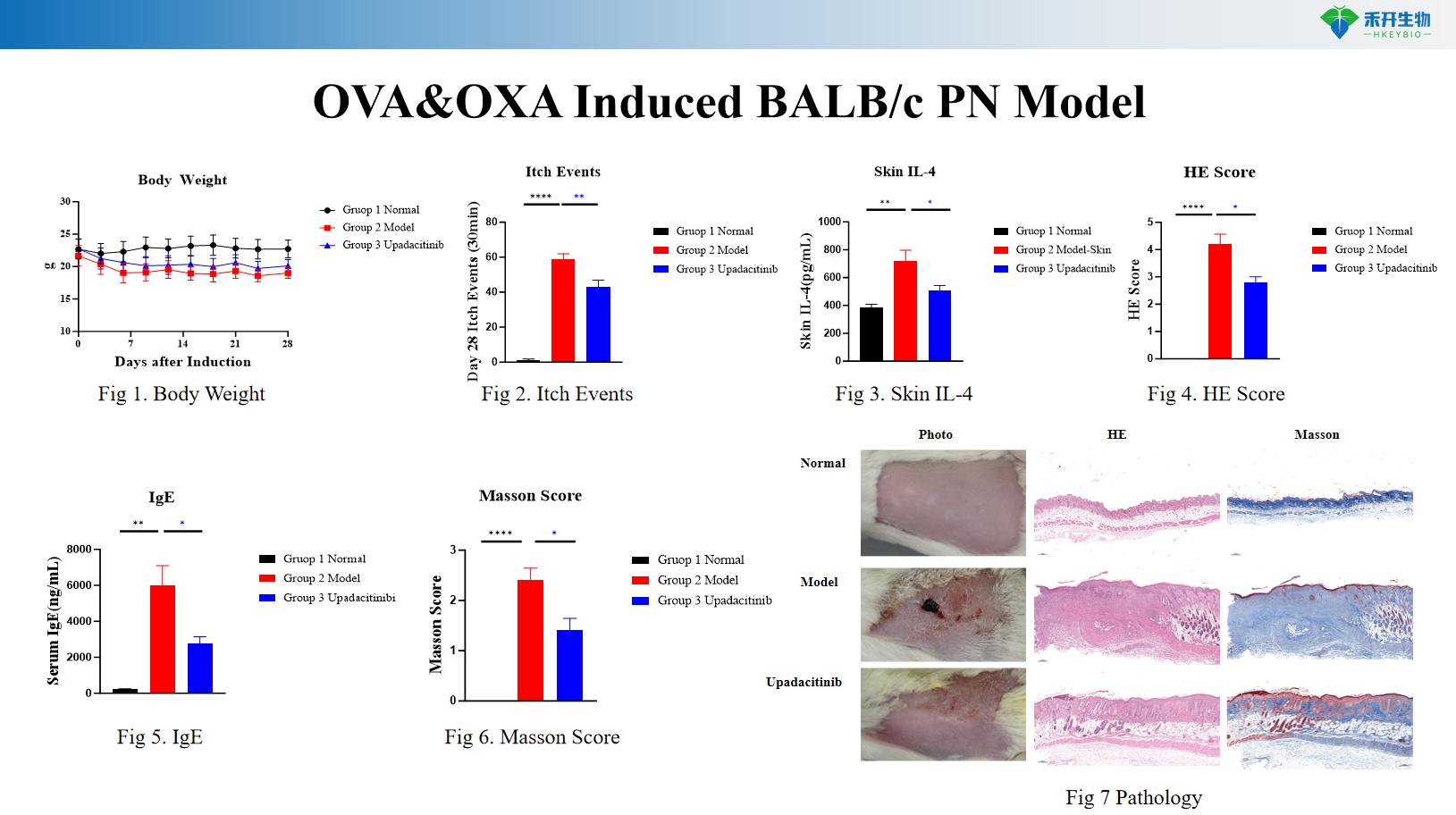
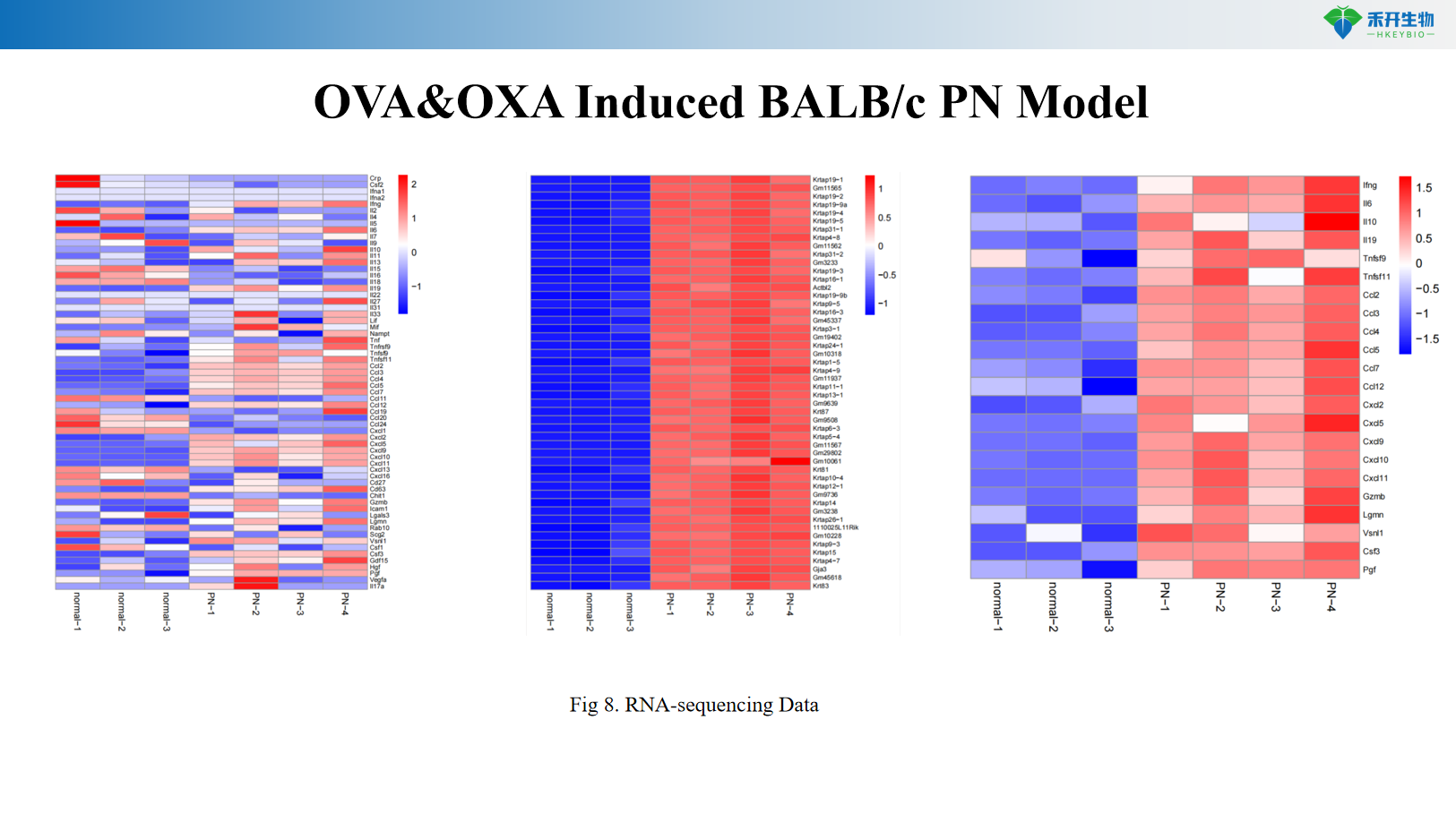
Composite endpoint – body weight, itch events (scratching behavior), serum IgE, skin IL-4, histopathology (HE, Masson trichrome), RNA sequencing data.
Translational Value – Ideal for testing anti-IgE biologics, JAK inhibitors, IL-4/13 antagonists, and antipruritic agents.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
OVA&OXA induced BALB/c PN model
Application areas
• Efficacy testing of anti-IgE biologics (omalizumab), JAK inhibitors (tofacitinib, upadacitinib), and IL-4/13 antagonists (dupilumab)
• Evaluation of antipruritic medications (antihistamines, neurokinin-1 receptor antagonists)
• Target validation of IgE-mediated chronic itch and fibrosis pathways
• Biomarker discovery (IgE, IL-4, pruritus-related mediators)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | BALB/c mouse |
induction method | OVA sensitization (intraperitoneal, with alum) + repeated topical OXA challenge on shaved back skin |
study time | 4–8 weeks (sensitization + challenge phase) |
critical endpoint | Body weight, pruritus events (scratching behavior), serum IgE (ELISA), skin IL-4 levels (ELISA), histopathology (HE score for epidermal hyperplasia and inflammation, Masson trichrome score for fibrosis), RNA sequencing data, optional: immunohistochemistry, flow cytometry |
| positive control | Corticosteroids (e.g. prednisolone) or anti-IgE antibodies may serve as reference compounds |
packet | Raw data, analysis reports, behavioral data, ELISA results, histological sections, RNA-seq data, bioinformatics (optional) |
❓ FAQ
Question: How do OVA and OXA induce prurigo nodularis in mice?
Answer: OVA sensitization can induce IgE and Th2 immunity. Repeated OXA challenge on the skin triggers a delayed-type hypersensitivity reaction that, combined with IgE-mediated inflammation, results in persistent scratching, papulondular lesion formation, and fibrosis, similar to the pathogenesis of human PN.
Q: What are the key similarities to prurigo nodularis in humans?
A: This model exhibits intense scratching, elevated IgE, Th2 cytokines (IL-4), epidermal hyperplasia, dermal fibrosis (Masson's trichrome), and papulonodular lesions that are closely related to human PN pathology.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g. different OVA/OXA doses, treatment times)?
Answer: Of course. Our scientific team tailors sensitization protocols, challenge plans, and endpoint analyzes for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Studies typically last 6-8 weeks and include sensitization, multiple OXA challenges, treatment, and endpoint analyses.





























