Key Features & Benefits
Clinically relevant – Recapitulates human PBC: AMA production, bile duct destruction, portal inflammation, and elevated inflammatory cytokines.
Mechanism-driven – Molecular mimicry between xenobiotic-modified 2OA and self-antigen lipoylated PDC-E2 breaks immune tolerance, inducing PBC-like pathology.
Comprehensive endpoints – Body weight, serum TNF-α and IFN-γ levels, liver histopathology (HE) with bile duct scoring, AMA detection (optional).
Translational value – Ideal for testing immunomodulators (corticosteroids, mycophenolate), bile acid therapies (UDCA), and biologics targeting inflammatory pathways.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
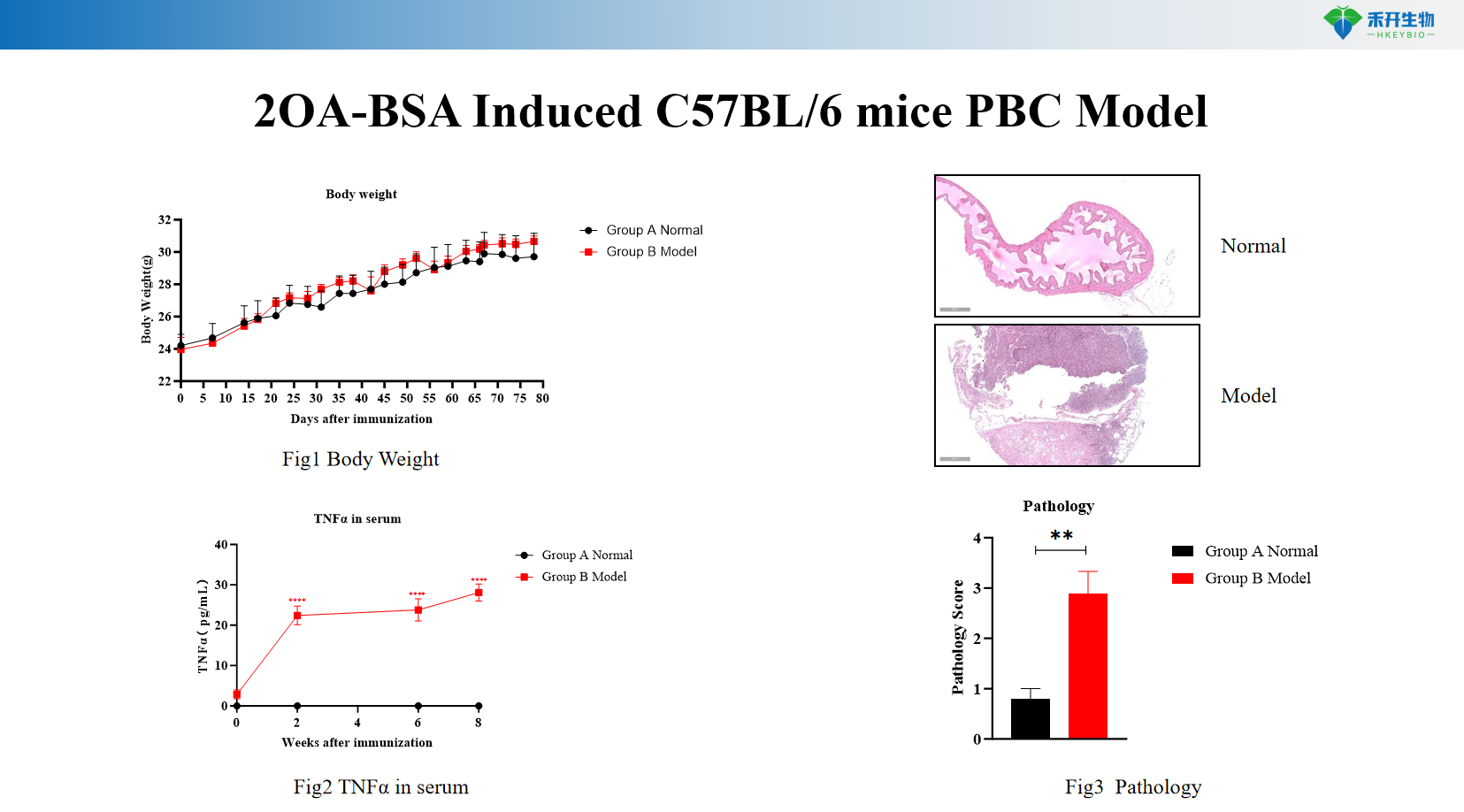
2OA-BSA Induced C57BL/6 mice PBC Model
Applications
• Efficacy testing of immunomodulators (corticosteroids, mycophenolate mofetil, azathioprine) for autoimmune cholangitis
• Evaluation of bile acid therapies (ursodeoxycholic acid, obeticholic acid) and anti-cholestatic agents
• Target validation for molecular mimicry and autoimmunity pathways in PBC
• Biomarker discovery (AMA, cytokine signatures, bile duct damage markers)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | C57BL/6 mouse |
Induction method | Immunization with 2-octynoic acid conjugated to bovine serum albumin (2OA-BSA) emulsified in complete Freund's adjuvant (CFA), with booster injections |
Study duration | 8–12 weeks (immunization + treatment phase) |
Key endpoints | Body weight, serum TNF-α and IFN-γ levels (ELISA), liver histopathology (HE staining with bile duct inflammation scoring), optional: anti-mitochondrial antibody (AMA) titers, immunohistochemistry for CK19 (bile duct epithelium), serum alkaline phosphatase (ALP) |
Data package | Raw data, analysis reports, histology slides, ELISA results, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does 2OA-BSA induce PBC in mice?
A: 2OA (2-octynoic acid) is a xenobiotic that structurally mimics the lipoic acid moiety of the self-antigen PDC-E2. Immunization with 2OA-BSA breaks immune tolerance via molecular mimicry, leading to activation of autoreactive T cells and production of anti-mitochondrial antibodies (AMA), which target bile duct epithelial cells and cause PBC-like pathology.
Q: What are the key similarities with human PBC?
A: The model exhibits AMA production, bile duct inflammation, portal tract infiltration, elevated inflammatory cytokines (TNF-α, IFN-γ), and liver histopathology consistent with human PBC, making it a valuable tool for studying disease mechanisms and testing therapeutics.
Q: Do you offer customized study protocols (e.g., different immunization schedules, dosing regimens)?
A: Absolutely. Our scientific team tailors immunization protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Pilot studies typically run 8–12 weeks post-immunization, including induction, treatment, and endpoint analysis.


























