Key Features & Benefits
Clinically relevant – BLM-induced skin fibrosis recapitulates human SSc with dermal thickening, collagen accumulation, and autoantibody production.
Mechanism-driven – BLM induces DNA damage, oxidative stress, and inflammation, leading to fibroblast activation and excessive collagen deposition.
Comprehensive endpoints – Body weight, skin pull-up height (mm), hydroxyproline content (collagen quantification), histopathology (HE, Sirius Red), dermal thickness measurement.
Translational value – Ideal for testing anti-fibrotic agents (nintedanib, pirfenidone), TGF-β inhibitors, tyrosine kinase inhibitors, and immunomodulators.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
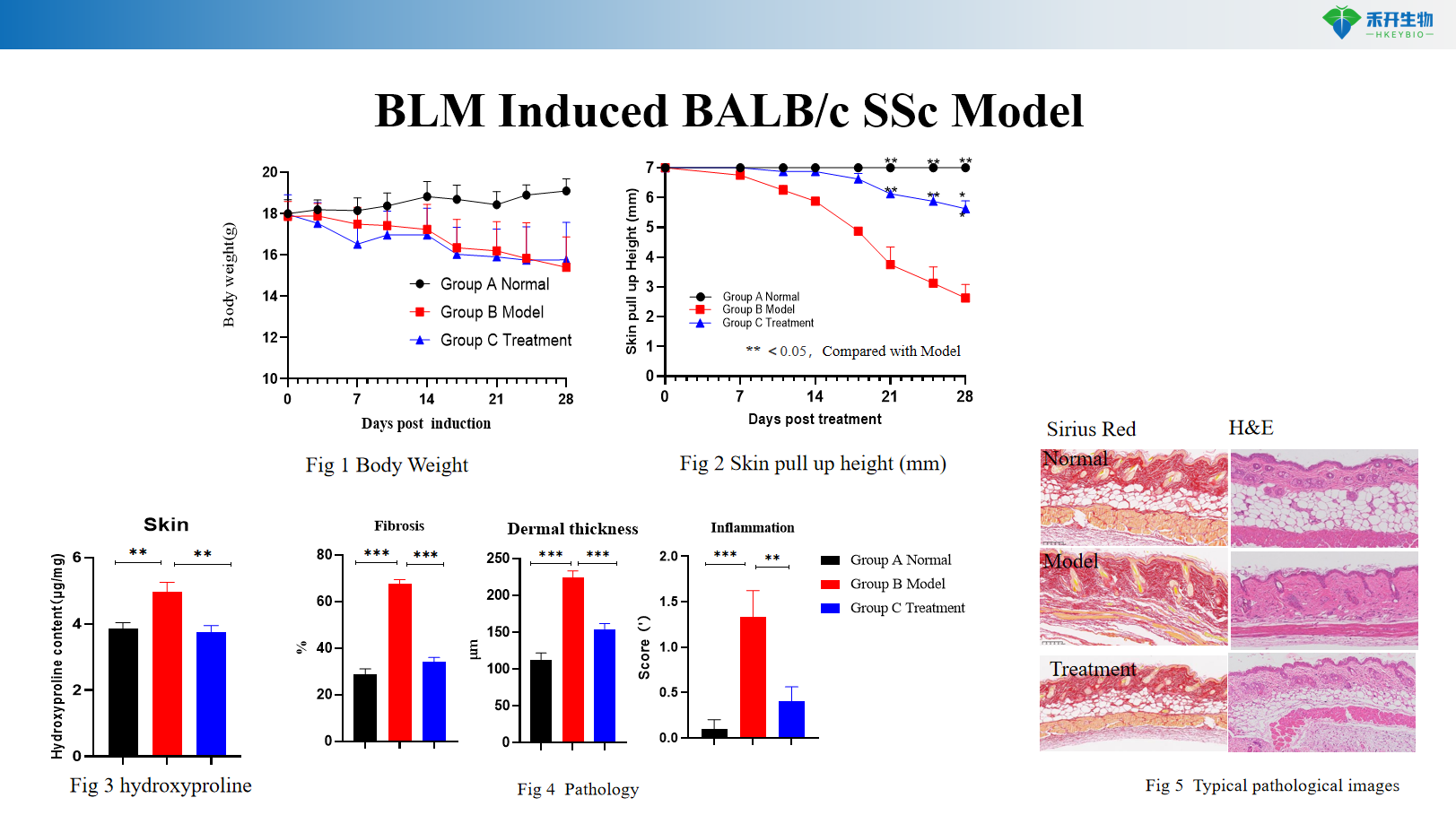
BLM Induced BALB/c SSc Model
Applications
• Efficacy testing of anti-fibrotic agents (nintedanib, pirfenidone, TGF-β inhibitors, galectin-3 inhibitors)
• Evaluation of tyrosine kinase inhibitors (imatinib, dasatinib) and immunomodulators (corticosteroids, mycophenolate)
• Target validation for fibrosis pathways (collagen synthesis, fibroblast activation)
• Biomarker discovery (hydroxyproline, collagen markers, inflammatory mediators)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | BALB/c mouse |
Induction method | Intradermal injection of bleomycin (BLM, 50–100 μL of 0.5–1 mg/mL solution) on shaved back skin, 2–3 times/week for 3–6 weeks |
Study duration | 3–8 weeks (induction + treatment phase) |
Key endpoints | Body weight, skin pull-up height (mm) as measure of skin thickness/fibrosis, hydroxyproline content (collagen quantification), skin histopathology (HE and Sirius Red staining with dermal thickness and collagen deposition scoring) |
| Positive control | Nintedanib or pirfenidone available as reference anti-fibrotic compounds |
| Data package | Raw data, analysis reports, histology slides (HE, Sirius Red), hydroxyproline assay results, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does bleomycin induce skin fibrosis?
A: Bleomycin causes DNA damage and oxidative stress in dermal fibroblasts and inflammatory cells, leading to activation of TGF-β signaling, fibroblast proliferation, and excessive collagen deposition, recapitulating human SSc fibrosis.
Q: What are the key similarities with human systemic sclerosis?
A: The model exhibits dermal thickening, increased collagen deposition, elevated hydroxyproline, and histopathological changes (HE, Sirius Red) that closely mimic human SSc skin fibrosis.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different BLM doses, injection sites, treatment schedules)?
A: Absolutely. Our scientific team tailors BLM dosing regimens, injection protocols, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Pilot studies typically run 4–6 weeks for fibrosis induction, with treatment initiated concurrently or after disease onset.

























