Key Features & Benefits
Clinically relevant – Recapitulates human asthma pathophysiology: airway hyperresponsiveness, eosinophilic inflammation, Th2 cytokine elevation.
Multiple allergen options – A.Suum protein and HDM induced models available.
Comprehensive endpoints – Includes lung function (airway resistance), BALF cell counts, IgE/IgG1 levels, histopathology, and cytokine profiling.
IND-ready data packages – Studies can be conducted in accordance with GLP rinciples.
Customizable protocols – Sensitization regimens, challenge schedules, and endpoint analyses tailored to your drug candidate.
Technical Data & Validation
Representative data from our NHP Asthma Model:
A.Suum Induced NHP Asthma Model
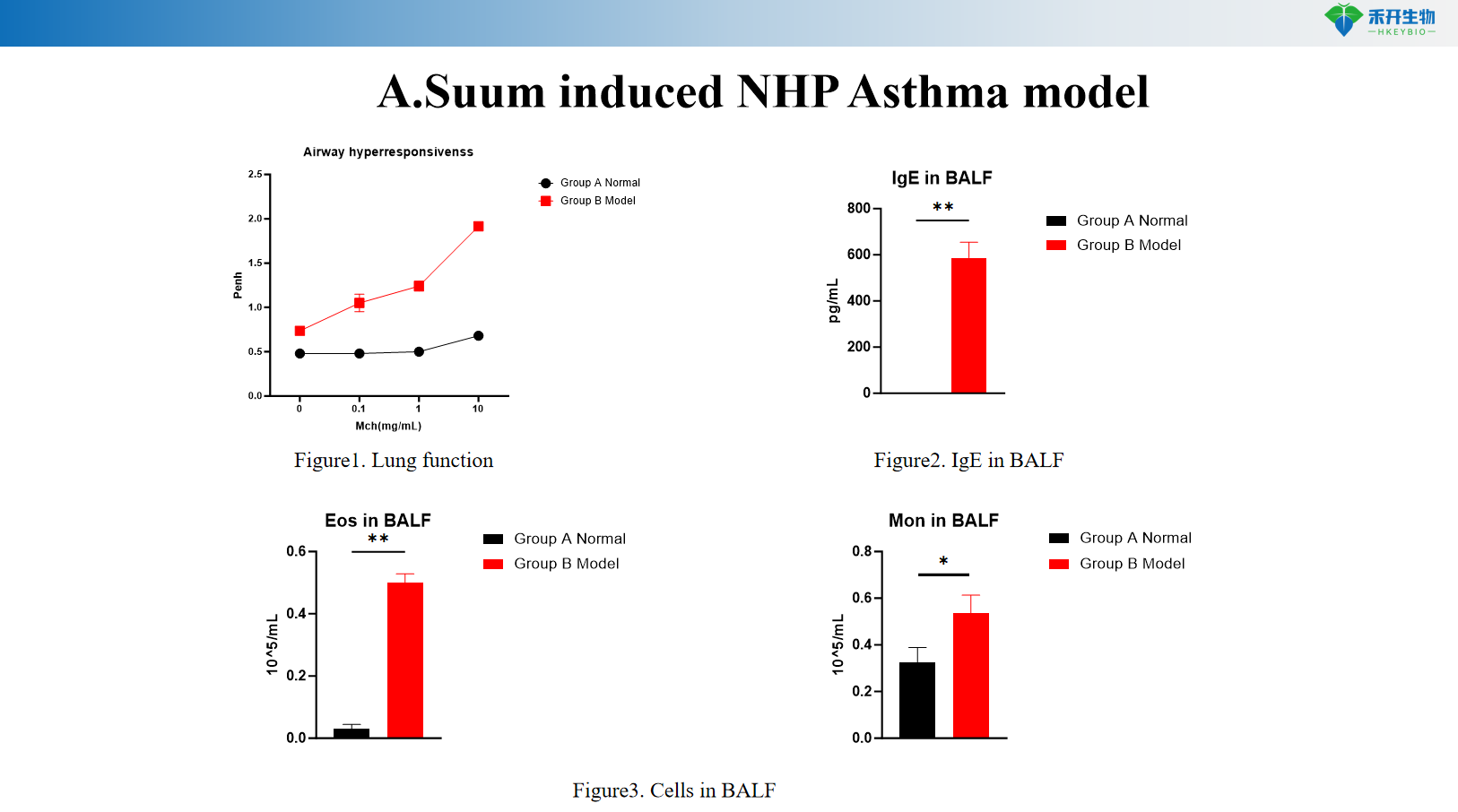
•
HDM induced NHP Asthma model
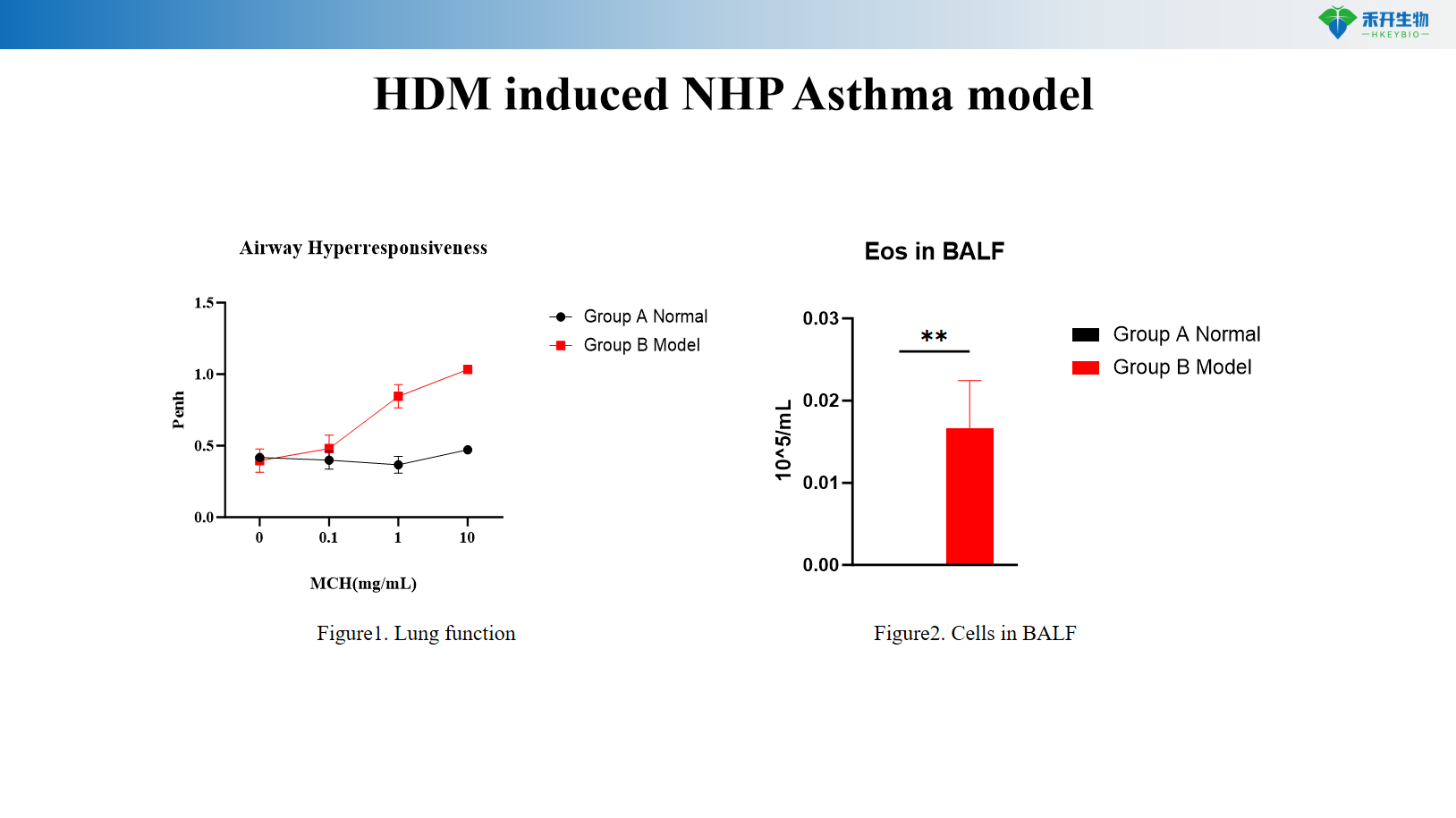
Applications
•Efficacy testing of asthma therapeutics (biologics, small molecules, inhaled drugs)
•Target validation for Th2-driven inflammation
•Biomarker discovery and validation (eosinophils, IgE, cytokines)
•IND-enabling toxicology and safety pharmacology studies
•Mechanism of action (MOA) investigations
Model Specifications
Parameter | A.Suum Induced Model | HDM Induced Model |
Species | Cynomolgus macaque (Macaca fascicularis) | Cynomolgus macaque (Macaca fascicularis) |
Induction method | A.Suum protein + Alum adjuvant (systemic sensitization) + aerosol challenge with A.Suum | House Dust Mite (HDM) extract, intratracheal or aerosol sensitization and challenge |
Study duration | 8–12 weeks | 6–10 weeks |
Key endpoints | Airway hyperresponsiveness, BALF eosinophils, IgE, IL-4/5/13, lung histopathology | Airway hyperresponsiveness, BALF eosinophils, total and HDM-specific IgE, Th2 cytokines, lung histopathology |
Data package | Raw data, analysis reports, histology slides, bioinformatics (optional) |
|
❓ Frequently Asked Questions
Q: How are the NHP asthma models induced?
A: Two models are available: A.Suum protein with alum adjuvant (systemic sensitization followed by aerosol challenge) and HDM (intratracheal or aerosol sensitization/challenge). Both induce eosinophilic airway inflammation and hyperresponsiveness.
Q: Can these models be used for IND-enabling studies?
A: Yes. We can perform studies in accordance with GLP principles; data packages are designed to support regulatory submissions (FDA, EMA).
Q:Do you offer customized study protocols?
A: Absolutely. Our scientific team tailors sensitization regimens, dosing schedules, and endpoint analyses to your drug candidate and MOA.
Q: What is the typical timeline for a pilot efficacy study?
A: Pilot studies (n=4-6/group) can be completed in 10–12 weeks, including sensitization, challenge, treatment, and endpoint analysis.

















