Main features and benefits
Clinical Relevance - The spontaneous autoimmune model closely resembles human autoimmune neuropathy, with progressive sensory deficits and pain.
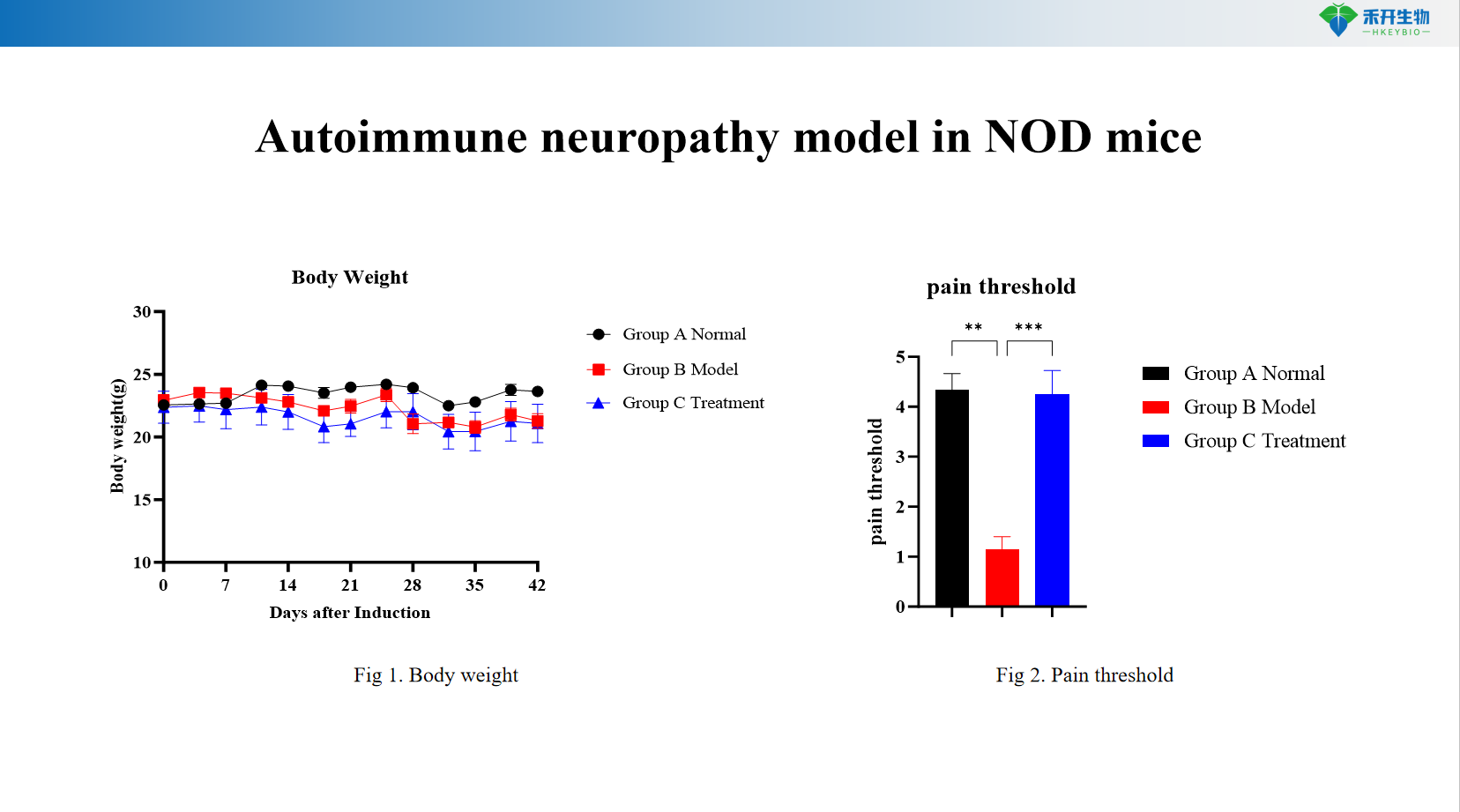
Quantifiable endpoints – weight monitoring, pain threshold measurement (von Frese), sensory function testing.
Mechanism-driven —T cell- and autoantibody-mediated autoimmune attack on peripheral nerves, reflecting the pathogenesis of human disease.
Translational value – ideal for testing immunomodulators (corticosteroids, IVIG, rituximab), analgesics (gabapentin, pregabalin) and neuroprotective agents.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
NOD mouse autoimmune neuropathy model
Application areas
• Testing the efficacy of immunomodulators (corticosteroids, IVIG, rituximab, mycophenolate mofetil) in autoimmune neuropathies
• Evaluation of treatments for neuropathic pain (gabapentin, pregabalin, tricyclic antidepressants, SNRI)
• Target validation of autoimmune-mediated neural injury and pain pathways
• Biomarker discovery (autoantibodies, cytokine profiles, pain-related mediators)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | Nord mouse |
induction method | Spontaneous autoimmune development (genetic predisposition); usually onset with advancing age |
study time | 8-20 weeks (monitoring from pre-onset to confirmed disease) |
critical endpoint | Body weight, pain threshold (Von Frey mechanical allodynia), sensory function tests (hot plate, cold sensitivity), optional: nerve conduction velocity, peripheral nerve histopathology (axonal degeneration, demyelination), immunohistochemistry (CD3, CD68, Iba1), serum autoantibodies (anti-neuronal antibodies) |
packet | Raw data, analysis report, behavioral data, histology slides, bioinformatics (optional) |
❓ FAQ
Q: How do NOD mouse models develop autoimmune neuropathy?
A: NOD mice have a genetic predisposition to autoimmune disease. They spontaneously generate an immune response against peripheral nerve components, leading to T-cell and autoantibody-mediated axonal and myelin damage, resulting in progressive sensory deficits and pain.
Q: What are the key similarities to human autoimmune neuropathies?
A: This model exhibits progressive weight loss, mechanical allodynia, sensory dysfunction, and histopathological evidence of neuroinflammation and damage, very similar to human autoimmune peripheral neuropathy.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different starting ages, combinations with pain medications)?
Answer: Of course. Our scientific team tailors monitoring plans, treatment options and endpoint analyzes for your specific drug candidate.































