Key Features & Benefits
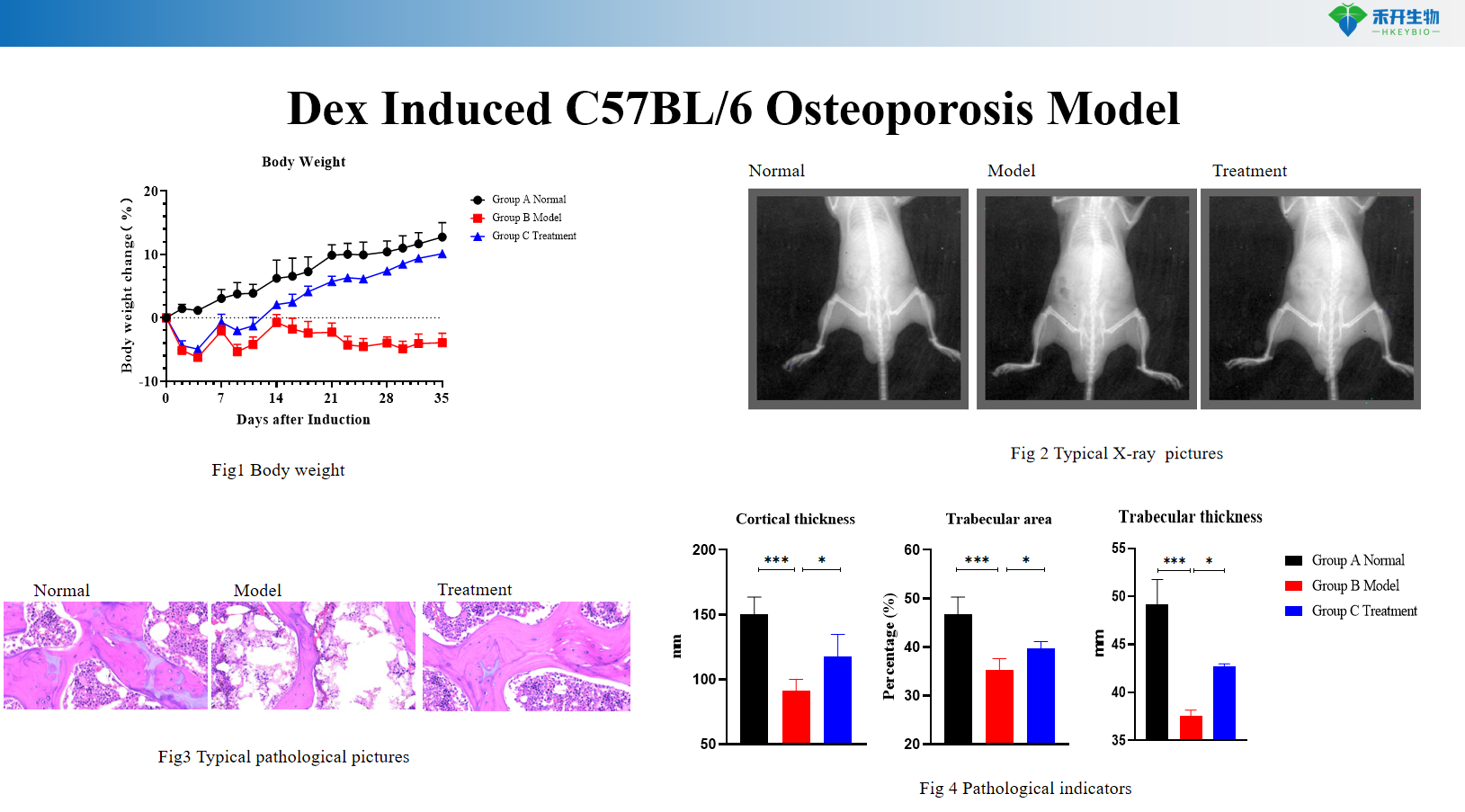
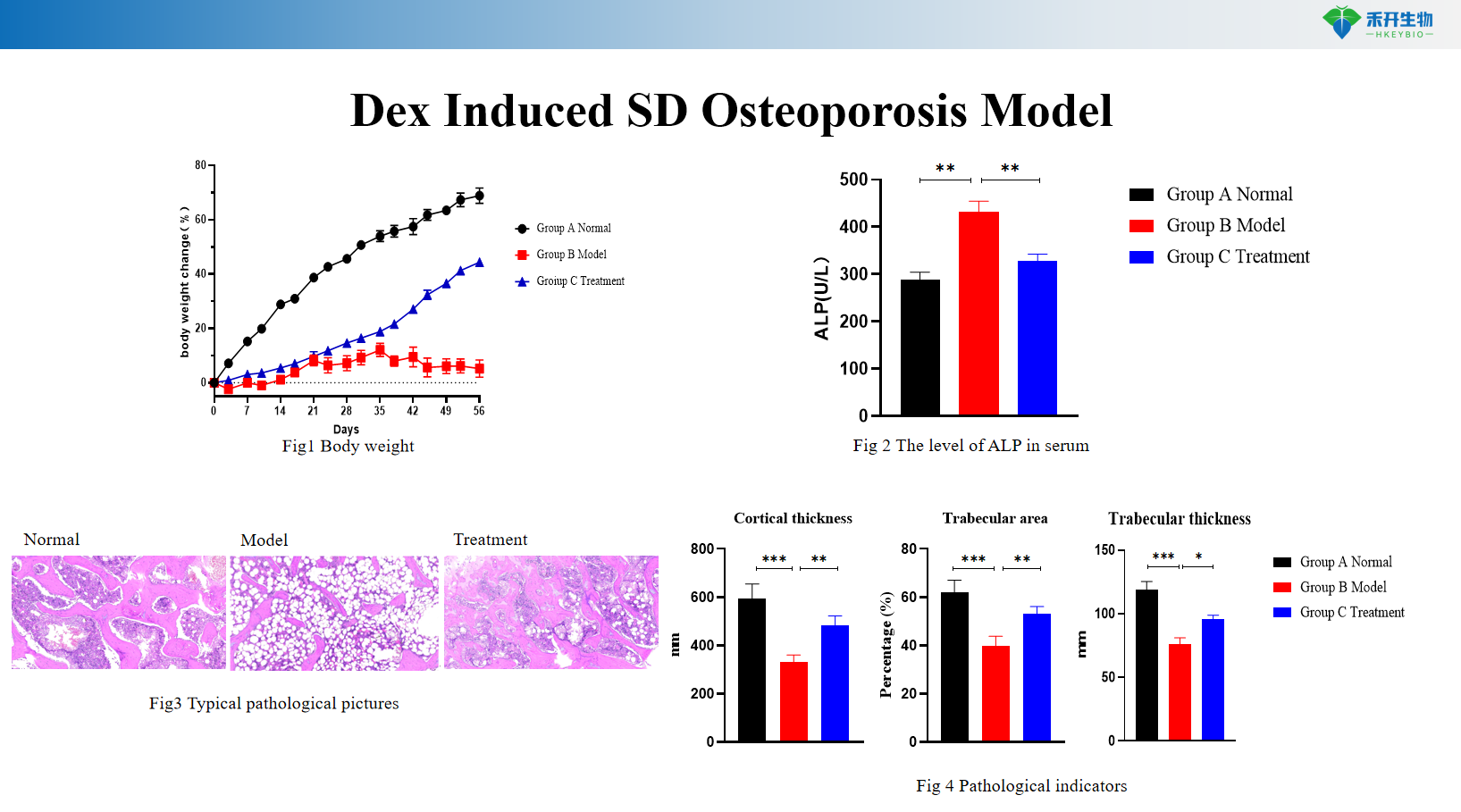
Clinically relevant – Recapitulates human glucocorticoid-induced osteoporosis with suppressed bone formation, enhanced resorption, and microarchitectural deterioration.
Multiple species – Rat and mouse models available to suit different experimental needs (larger size vs. genetic manipulation).
Comprehensive endpoints – Body weight, serum ALP, X-ray imaging, histopathology (H&E, TRAP staining), bone histomorphometry, micro-CT (optional).
Translational value – Ideal for testing bisphosphonates (alendronate), SERMs (raloxifene), anabolic agents (teriparatide), and RANKL inhibitors.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Dex Induced C57BL/6 Osteoporosis Model
Dex Induced SD Osteoporosis Model
Applications
• Efficacy testing of anti-osteoporotic agents: bisphosphonates (alendronate, zoledronic acid), SERMs (raloxifene), anabolic agents (teriparatide, romosozumab), and RANKL inhibitors (denosumab)
• Evaluation of novel therapies targeting bone formation or resorption pathways
• Target validation for glucocorticoid-induced osteoporosis mechanisms
• Biomarker discovery (bone turnover markers, ALP, osteocalcin, CTX-I)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | SD Rat OP Model | C57BL/6 Mouse OP Model |
Species/Strain | Sprague-Dawley rat | C57BL/6 mouse |
Induction method | Dexamethasone (1–2.5 mg/kg) administered intramuscularly or subcutaneously, 2–3 times/week for 4–8 weeks |
Study duration | 4–10 weeks (induction + treatment phase) |
Key endpoints | Body weight, serum ALP, X-ray imaging, histopathology (H&E, TRAP), bone histomorphometry, micro-CT (optional) | Body weight, serum ALP, histopathology, micro-CT (optional) |
| Positive control | Alendronate or teriparatide available as reference compounds |
Data package | Raw data, analysis reports, X-ray images, histology slides, micro-CT data (optional), bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does dexamethasone induce osteoporosis in rodents?
A: Dexamethasone suppresses osteoblast differentiation and promotes osteoblast apoptosis, leading to reduced bone formation. It also indirectly enhances osteoclast activity and survival, resulting in net bone loss, thinning of trabeculae, and increased fracture risk, mimicking human glucocorticoid-induced osteoporosis.
Q: What are the key similarities with human osteoporosis?
A:Both models exhibit decreased bone mineral density, microarchitectural deterioration, elevated serum ALP, and histomorphometric changes (thinned trabeculae, marrow adiposity) that closely mirror human secondary osteoporosis.
Q: Can these models be used for IND-enabling studies?
A:Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different dexamethasone doses, treatment durations)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Pilot studies typically run 6–8 weeks, including dexamethasone induction and treatment phases, with endpoint analyses at week 8.

























