Key Features & Benefits
Broad model portfolio – Spontaneous (MRL/lpr), chemical (pristane), TLR-driven (imiquimod), antigen-driven (ALD-DNA, apoptotic cells), and humanized models.
Multiple strains – MRL/lpr, C57BL/6, BALB/c, and humanized mice available.
Comprehensive endpoints – Body weight, lymphadenopathy/spleen/kidney indices, anti-dsDNA, proteinuria, serum CREA/LDH/AST, renal histopathology (HE, IgG deposition), flow cytometry (B cells, plasma cells, T cells).
Translational value – Ideal for testing immunosuppressants, biologics (anti-CD20, anti-IFNAR), TLR inhibitors, and B cell‑targeted therapies.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
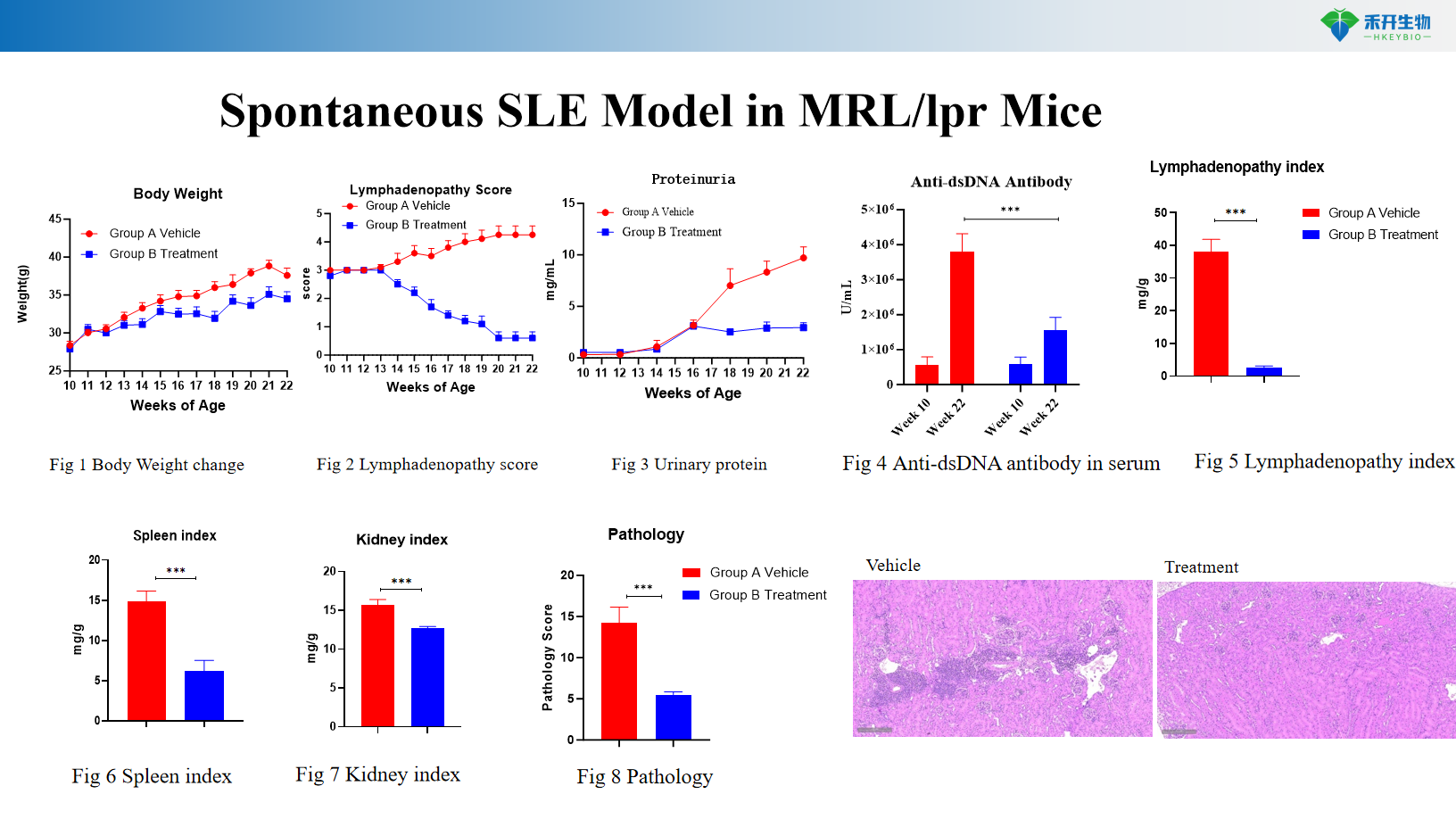
Spontaneous SLE Model in MRL/lpr Mice
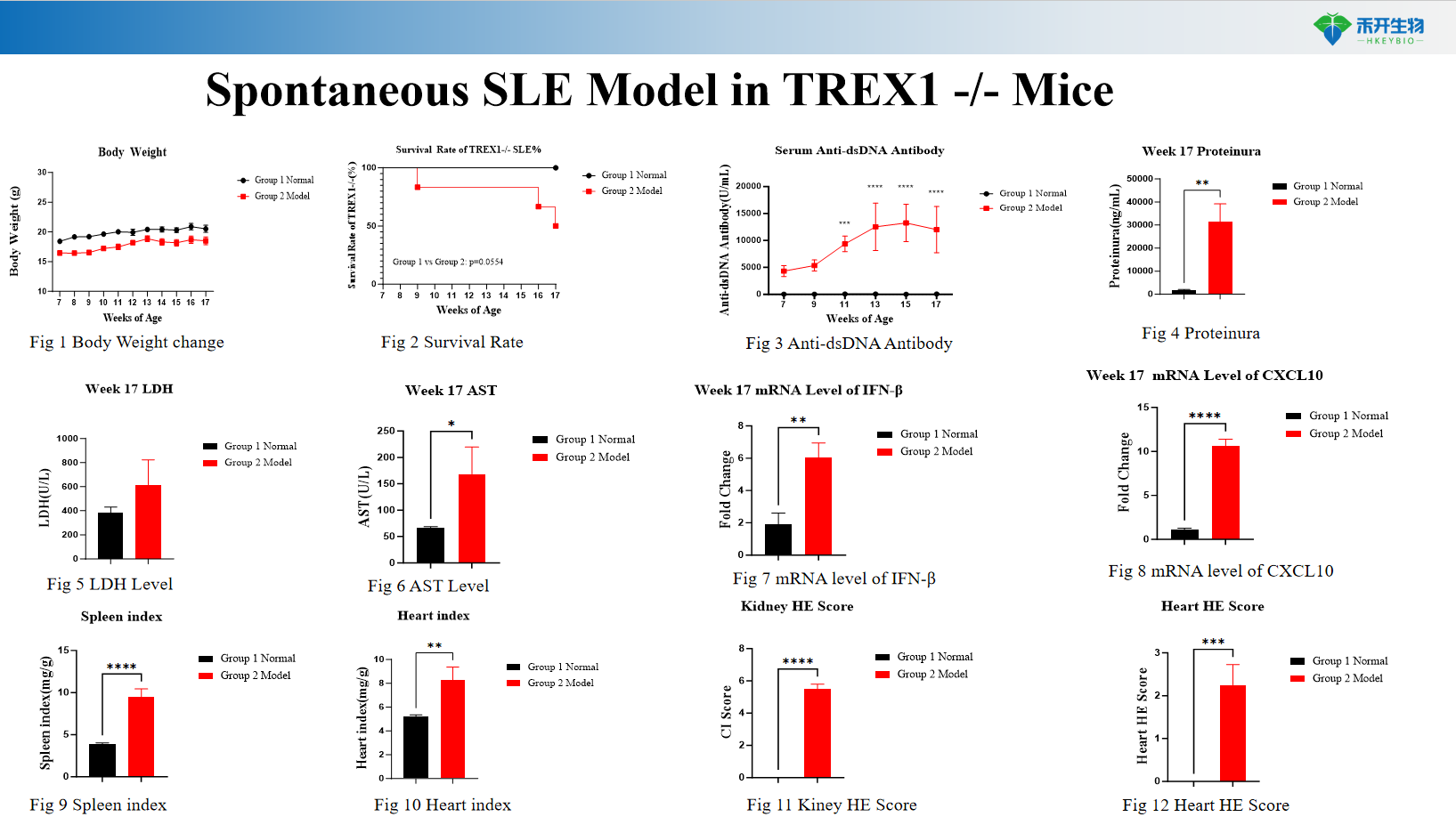
Spontaneous SLE Model in TREX1 -/- Mice
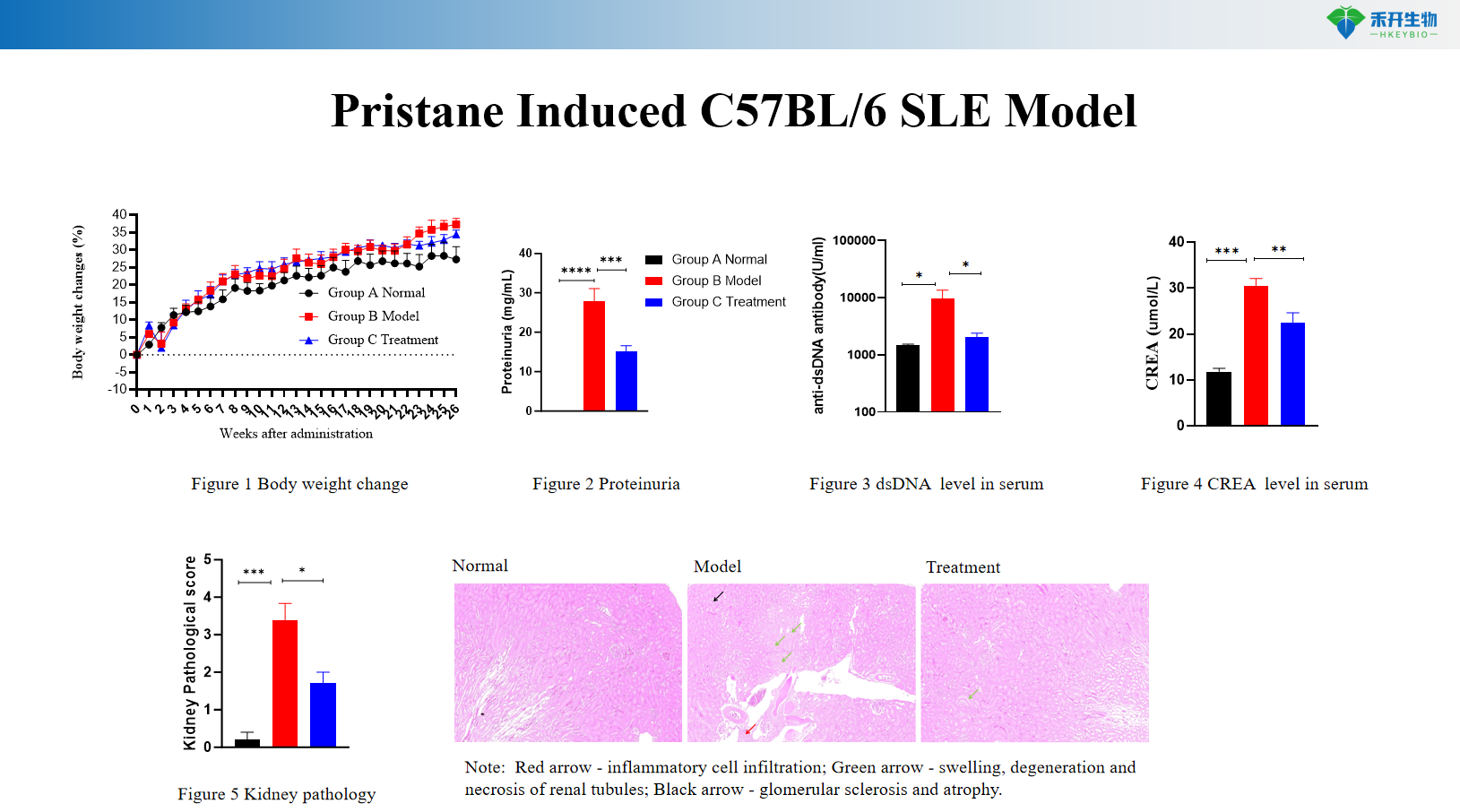
Pristane Induced C57BL/6 SLE Model
TLR-7 Agonist Induced SLE model in C57BL/6 Mice
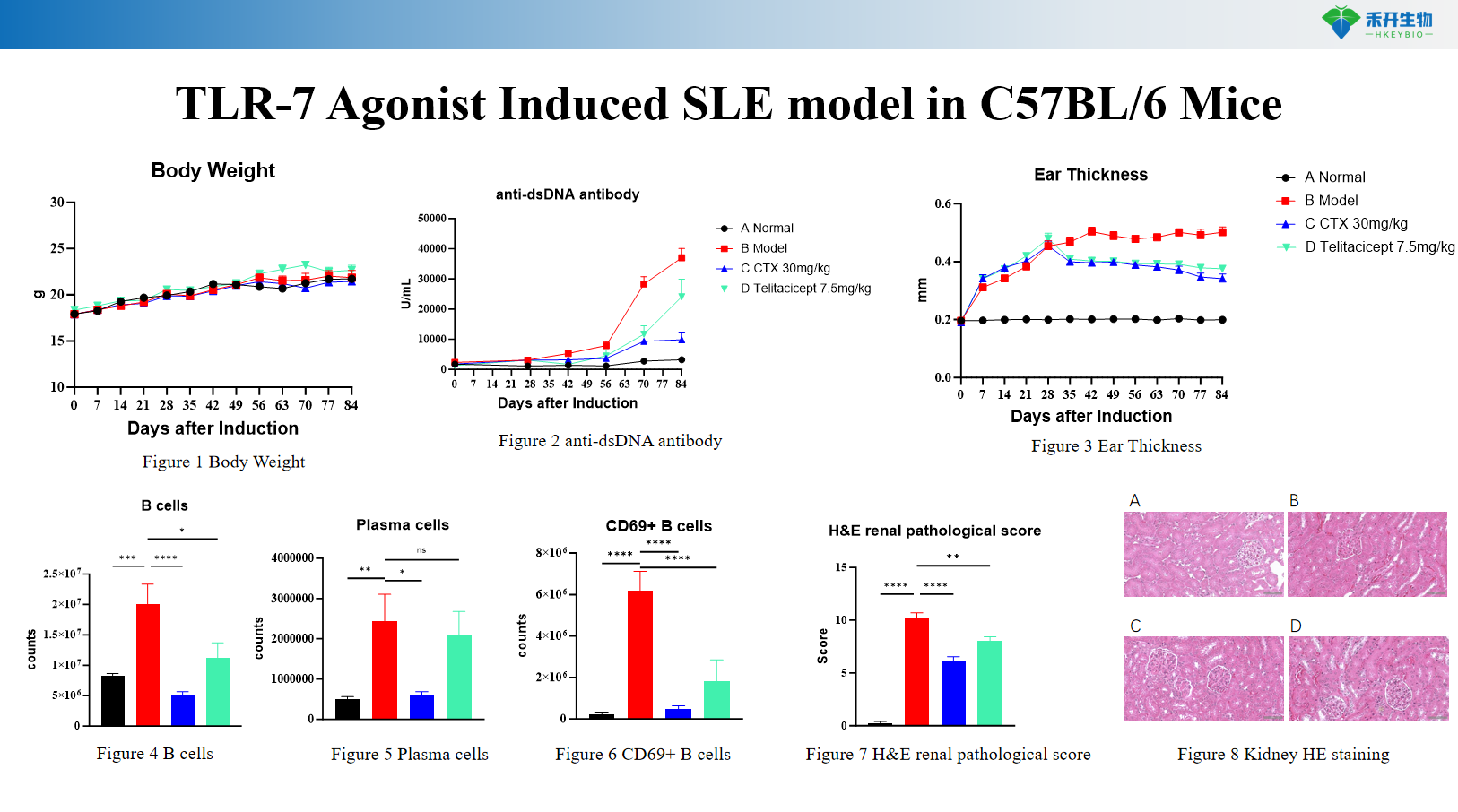
TLR-7 Agonist Induced SLE model in C57BL/6 Mice
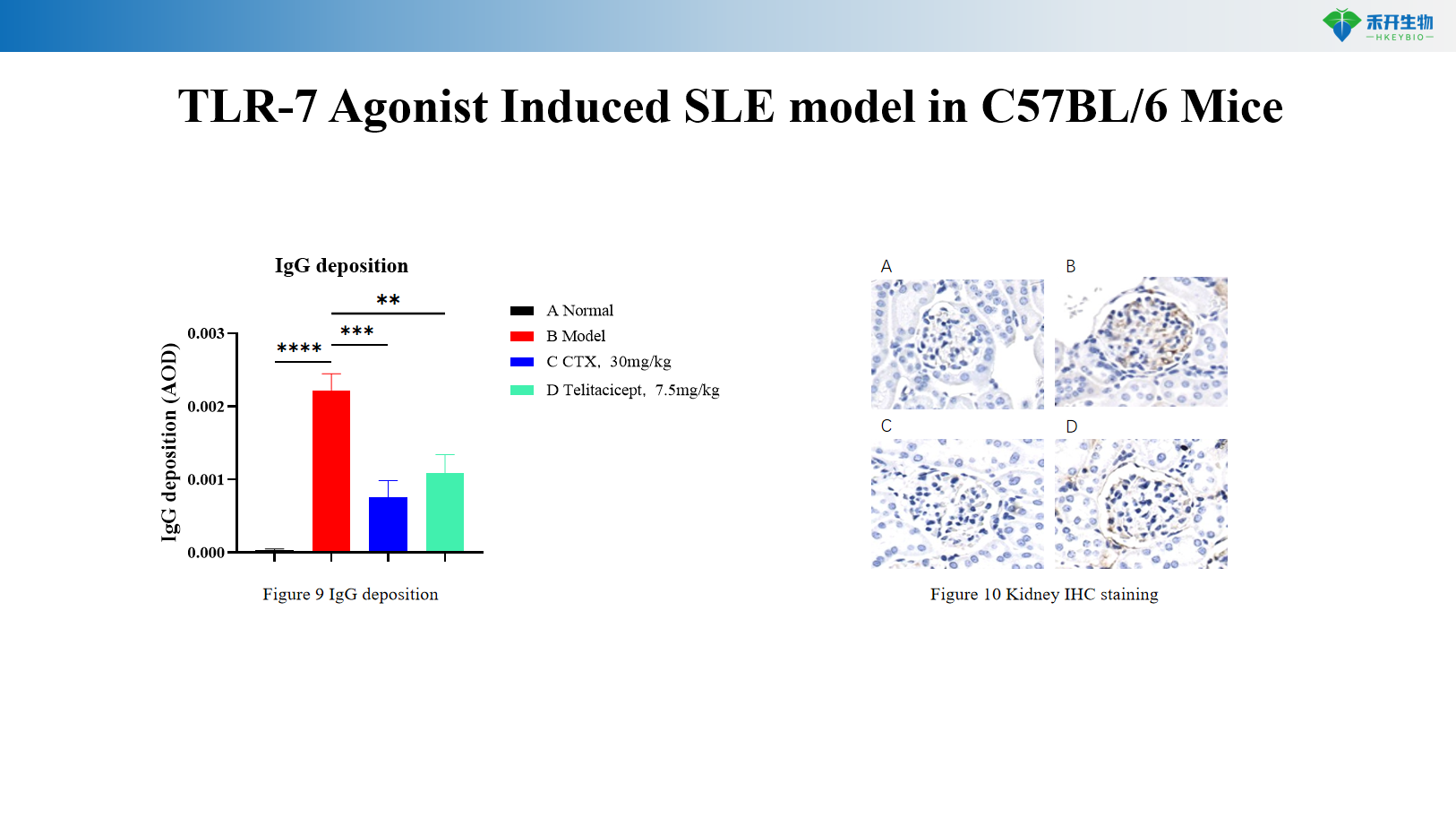
TLR agonist induced humanized SLE model
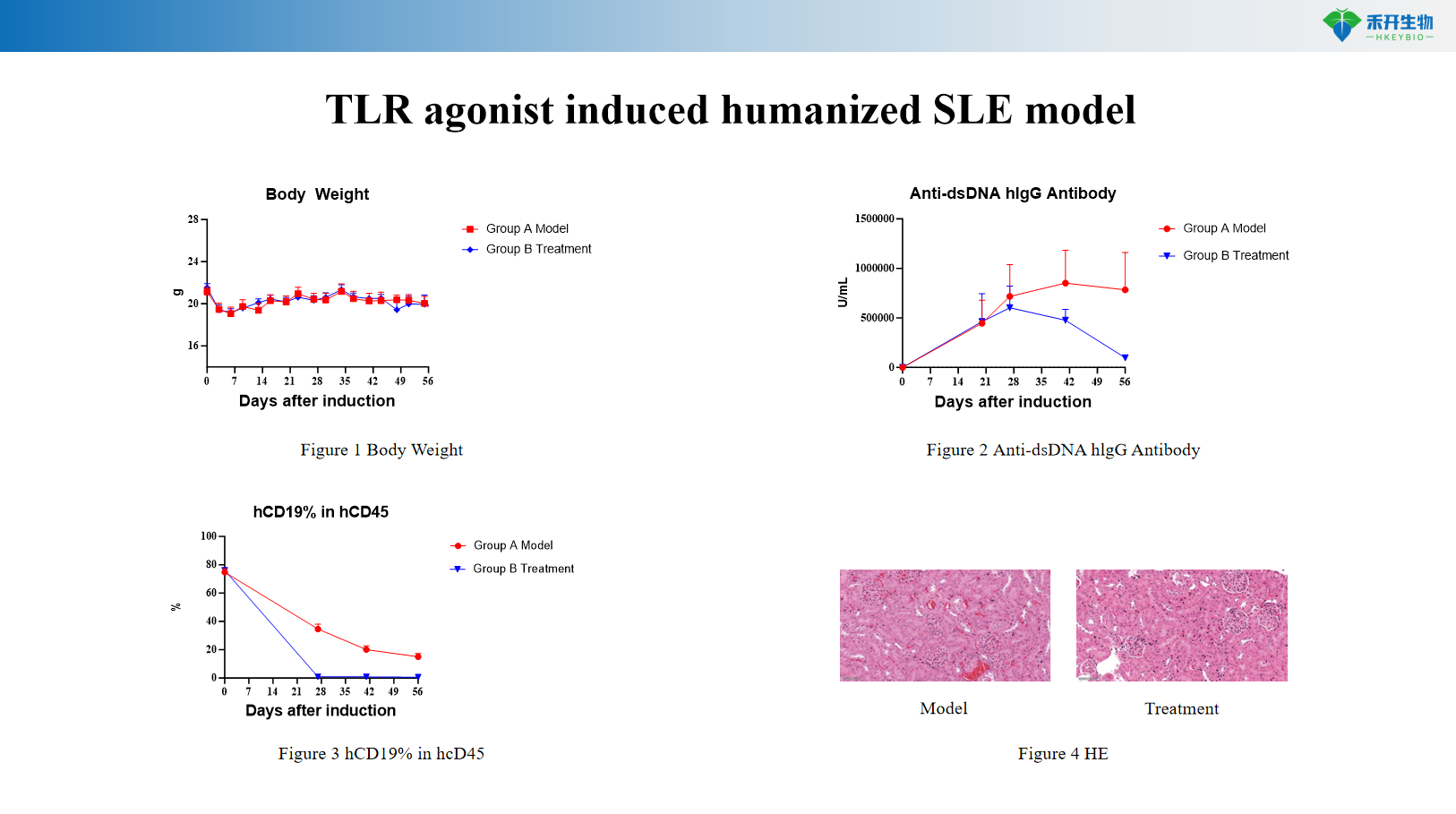
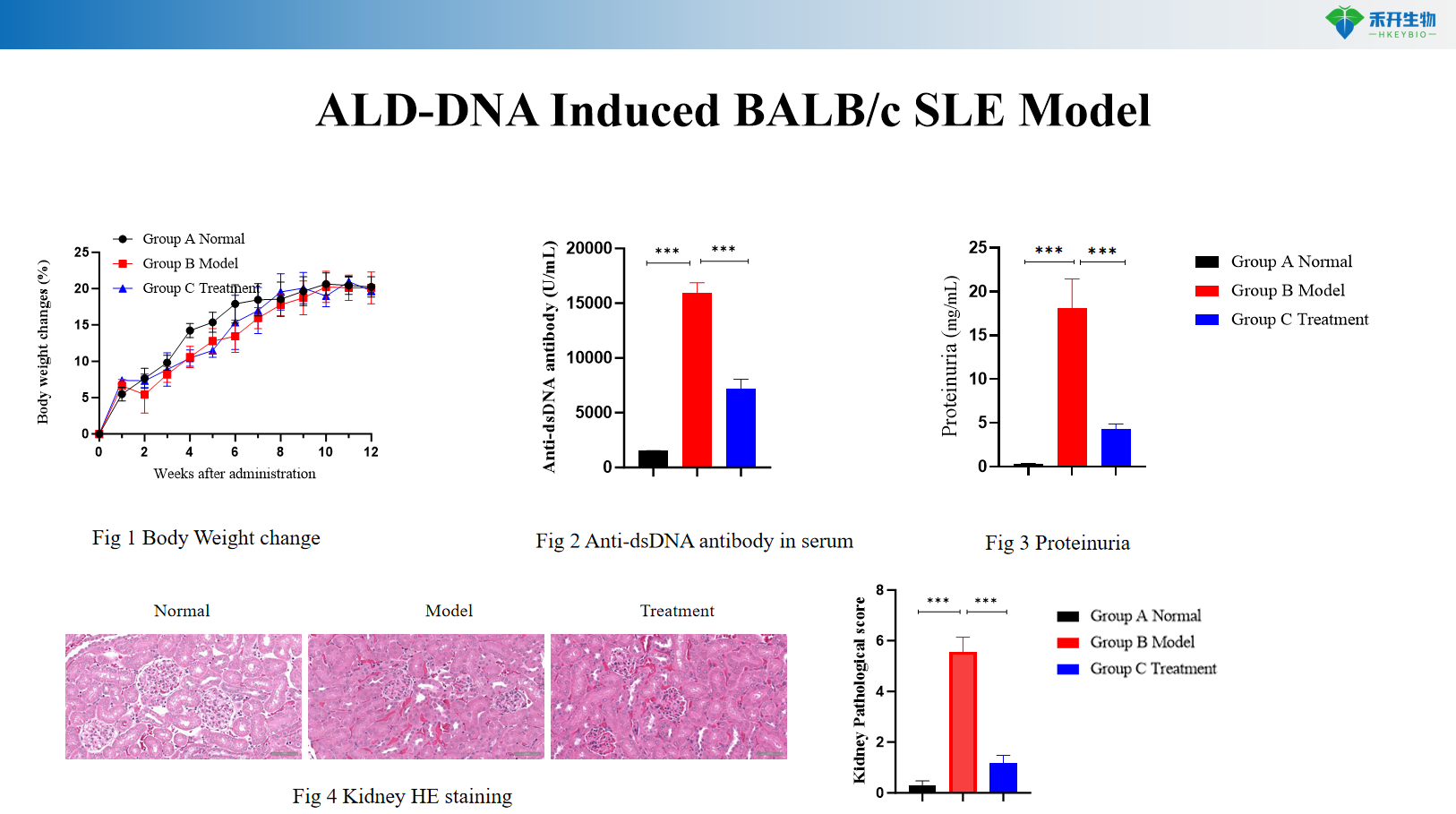
ALD-DNA Induced BALB/c SLE Model
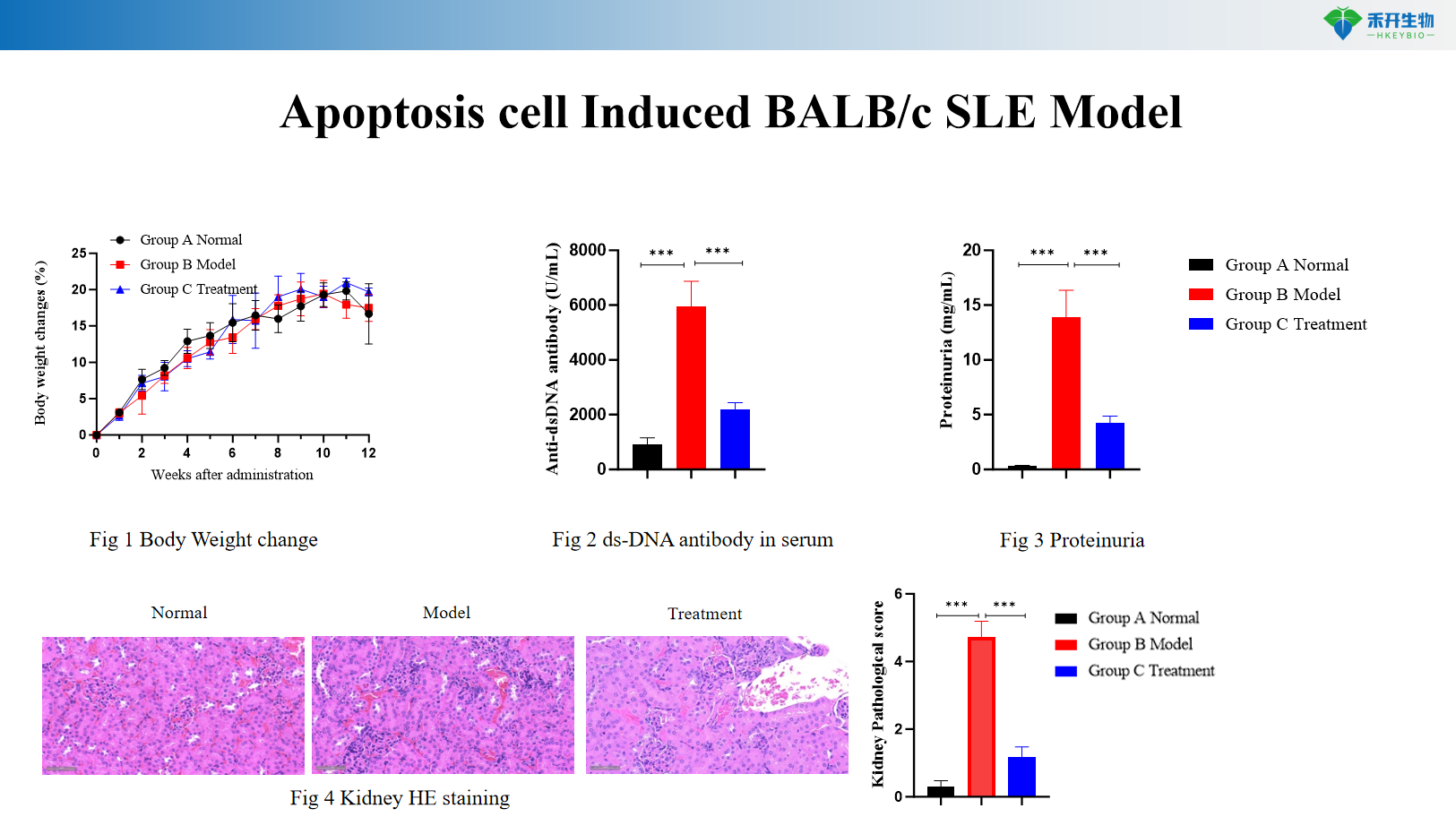
Apoptosis cell Induced BALB/c SLE Model
Applications
• Efficacy testing of immunosuppressants (cyclophosphamide, mycophenolate, corticosteroids) and biologics (anti-CD20, anti-BAFF, anti-IFNAR)
• Evaluation of TLR7/9 inhibitors, JAK inhibitors, and B cell‑targeted therapies
• Target validation for autoantibody production, type I interferon signature, and nephritis pathways
• Biomarker discovery (anti-dsDNA, proteinuria, cytokine signatures)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strains | MRL/lpr, C57BL/6, BALB/c, humanized mice |
Induction methods | Spontaneous (Fas mutation); pristane i.p.; topical imiquimod (TLR-7 agonist); immunization with ALD-DNA or apoptotic cells |
Study duration | Spontaneous: 12–20 weeks; induced: 4–16 weeks depending on model |
Key endpoints | Body weight, lymphadenopathy/spleen/kidney indices, serum anti-dsDNA antibodies, proteinuria, serum CREA/LDH/AST, renal histopathology (HE, IgG/IgM deposition), flow cytometry (B cells, plasma cells, T cells), type I interferon signature (ISG expression |
| Positive control | Cyclophosphamide or mycophenolate mofetil available as reference compounds |
| Data package | Raw data, analysis reports, clinical chemistry, histology slides, flow cytometry files, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What are the differences between spontaneous and induced SLE models?
A: Spontaneous models (MRL/lpr) develop disease naturally over time, mimicking chronic progressive SLE. Induced models (pristane, TLR-7, ALD-DNA) offer faster, synchronized onset and allow study of specific triggers. Humanized models enable evaluation of human-specific biologics.
Q: Which model is best for testing anti‑IFNAR biologics?
A: The pristane and TLR‑7 agonist models exhibit strong type I interferon signatures, making them suitable for evaluating anti‑IFNAR antibodies (e.g., anifrolumab).
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different induction doses, treatment timing)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Induced models: 4–16 weeks; spontaneous MRL/lpr: 12–20 weeks. Humanized models require additional time for immune reconstitution.































