Key Features & Benefits
Clinically relevant – Combined cigarette smoke and LPS exposure mimics human COPD etiology and chronic inflammation.
Multiple species – Mouse and rat models available to suit different experimental requirements (genetic tools vs. larger size).
Comprehensive endpoints – Body weight, BALF cytology (WBC, macrophages, monocytes), lung function (resistance, compliance), histopathology (HE scoring), cytokine profiling.
Translational value – Ideal for testing anti-inflammatory drugs (corticosteroids, PDE4 inhibitors), bronchodilators, and mucolytics.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
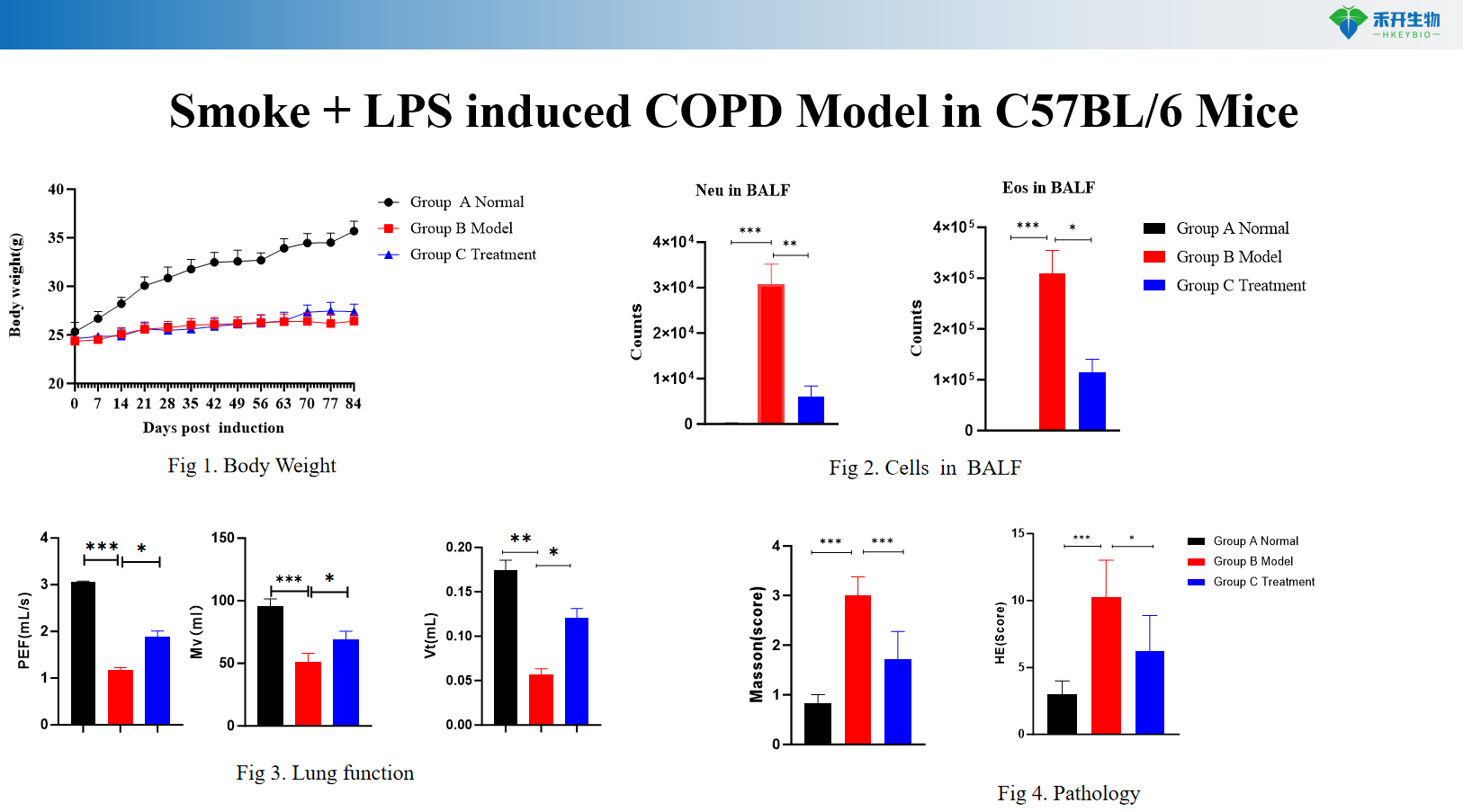
Smoke + LPS induced COPD Model in C57BL/6 Mice
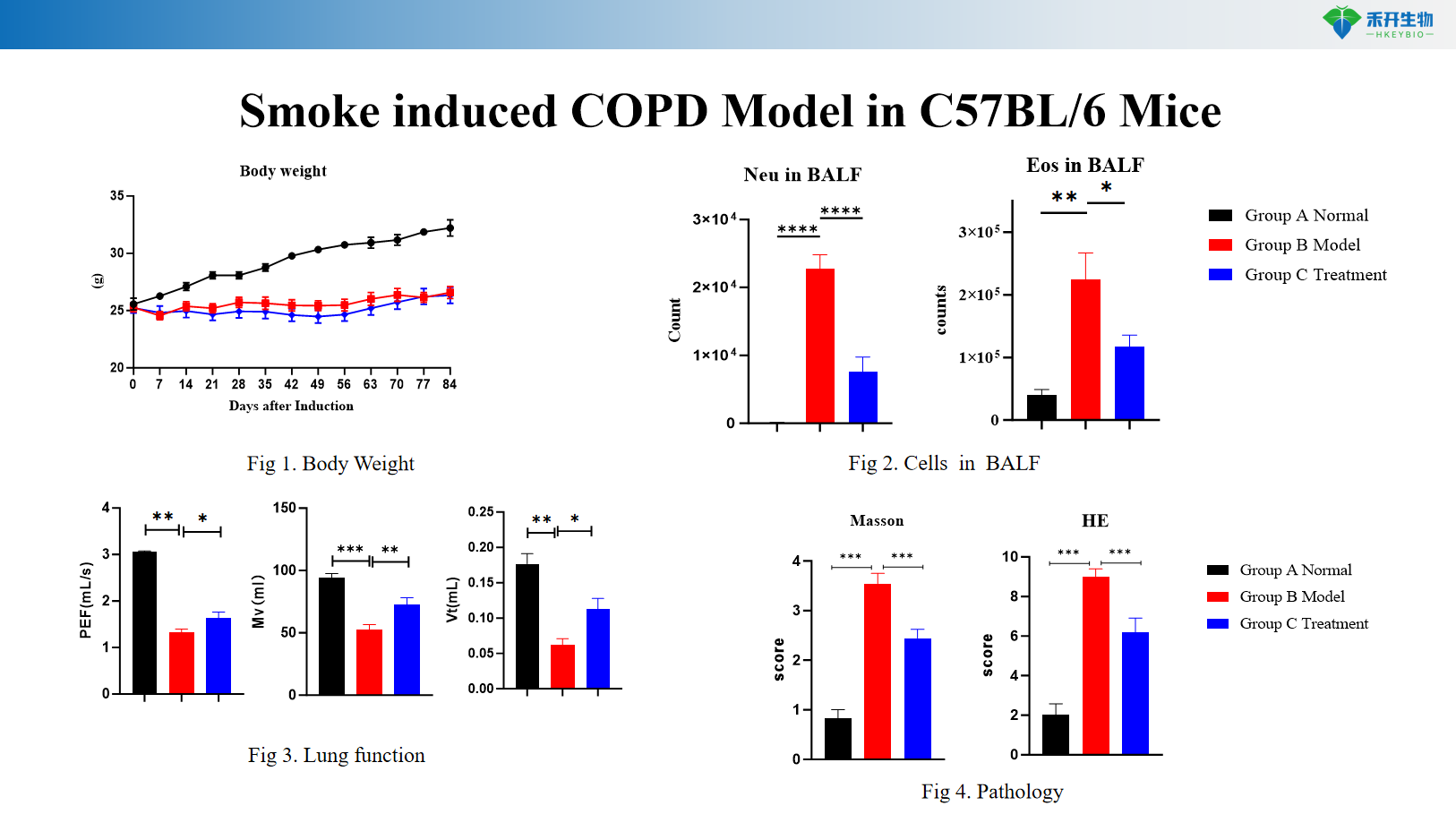
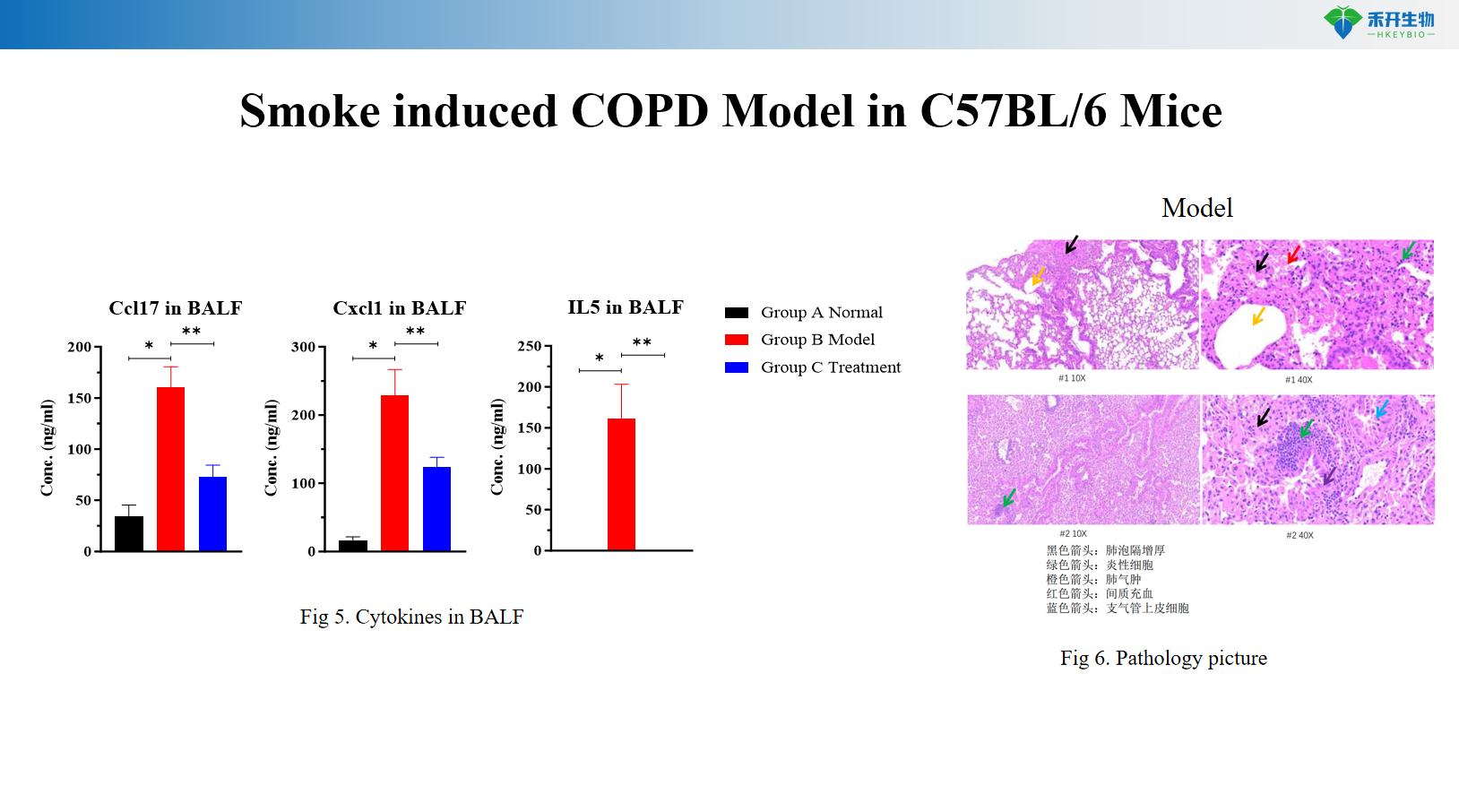
Smoke induced COPD Model in C57BL/6 Mice
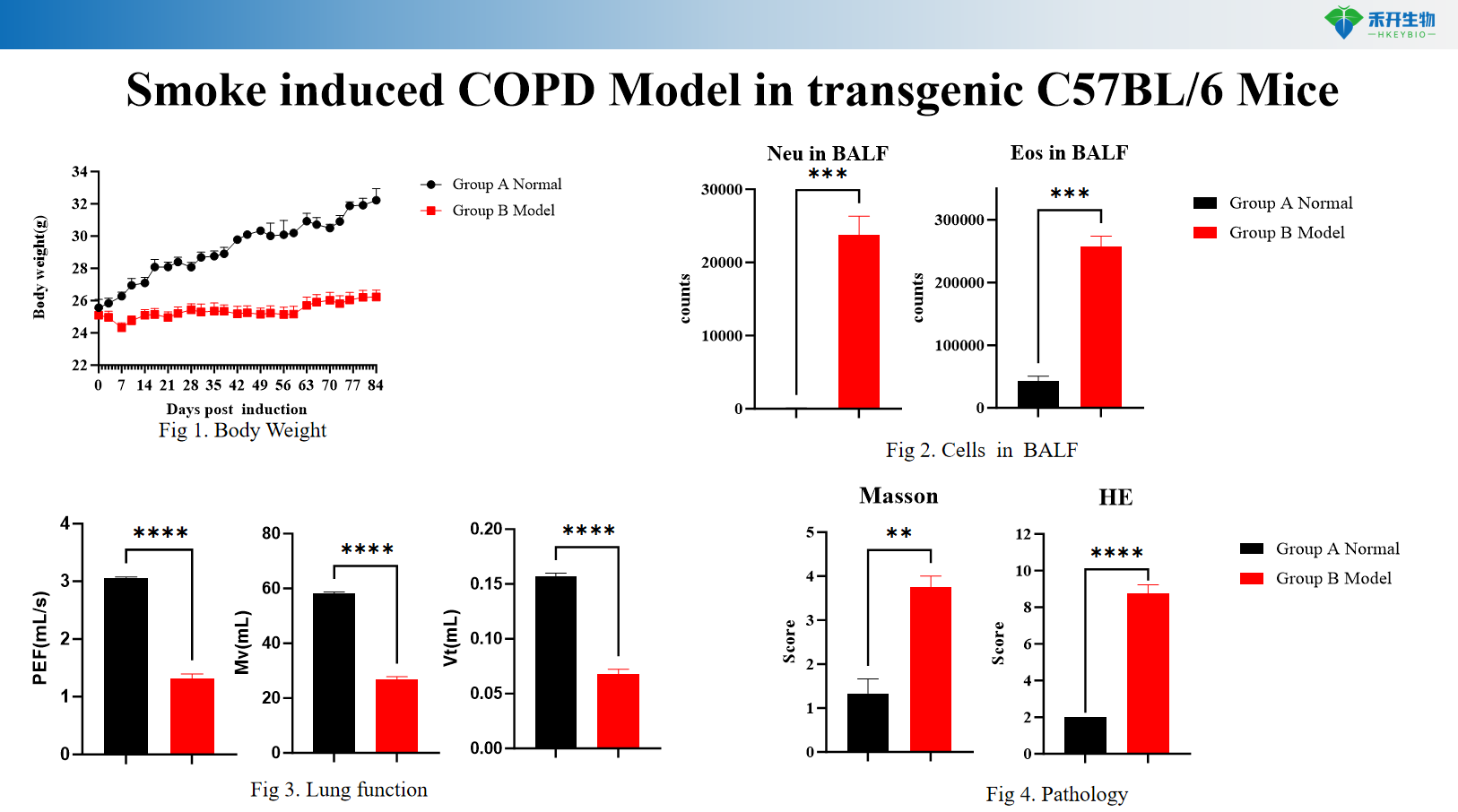
Smoke induced COPD Model in transgenic C57BL/6 Mice
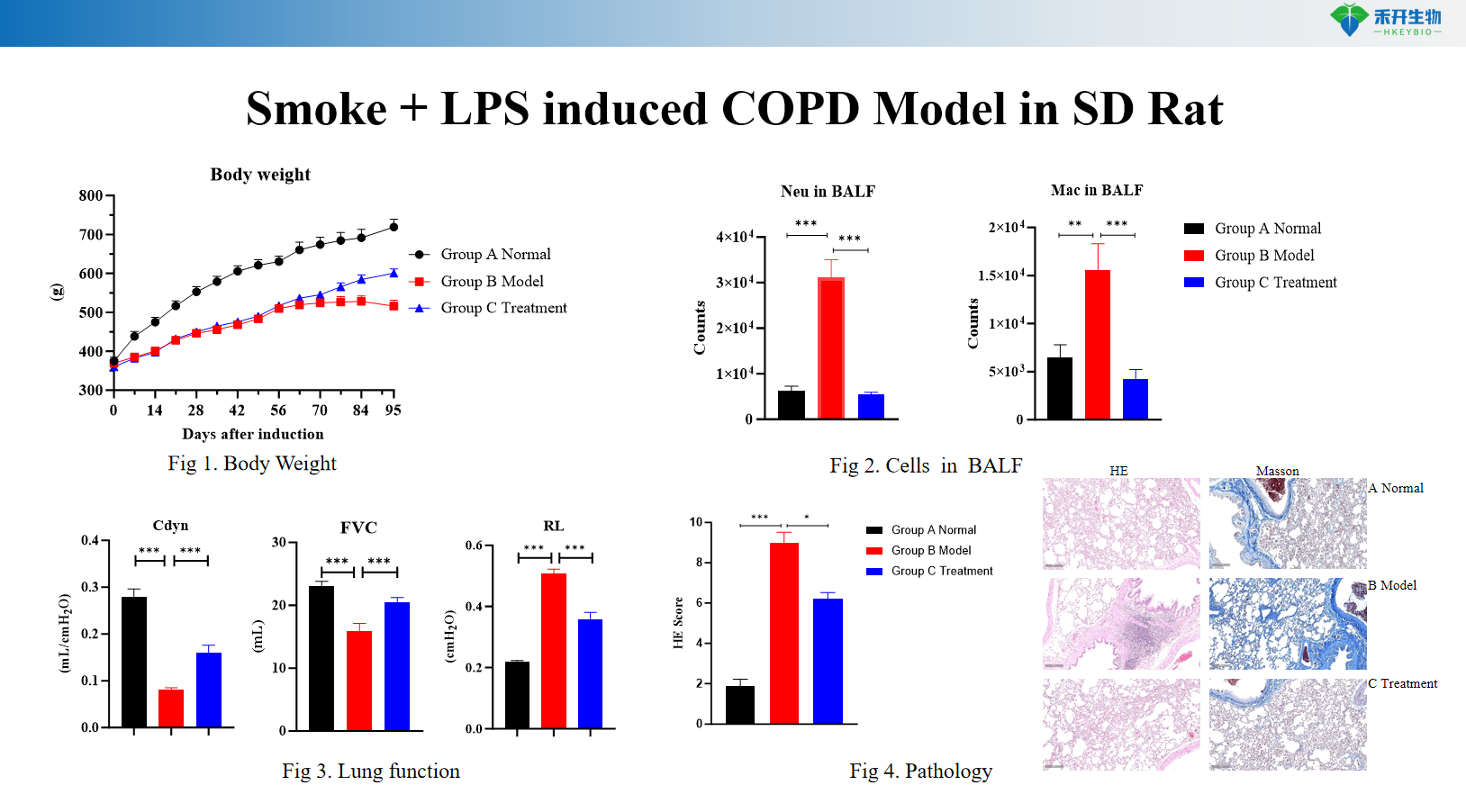
Smoke + LPS induced COPD Model in SD Rat
Applications
• Efficacy testing of anti-inflammatory drugs (corticosteroids, PDE4 inhibitors, p38 MAPK inhibitors)
• Evaluation of bronchodilators (β2-agonists, anticholinergics) and combination therapies
• Testing of mucoactive agents and antioxidants
• Target validation for inflammatory and oxidative stress pathways
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Mouse COPD Model | Rat COPD Model |
Species/Strain | C57BL/6 mouse | Sprague-Dawley rat |
Induction method | Chronic cigarette smoke exposure (whole-body or nose-only) 5 days/week for 4–6 months + intratracheal LPS (1–2 times/week) |
Study duration | 4–7 months (induction + treatment) | 4–7 months (induction + treatment) |
Key endpoints | Body weight, BALF cell counts (total and differential: macrophages, monocytes, neutrophils), lung function (resistance, compliance, Penh), histopathology (HE scoring for emphysema and inflammation), cytokine levels in BALF (IL-6, TNF-α, MCP-1), optional: oxidative stress markers, goblet cell metaplasia (PAS), mean linear intercept (MLI) |
Data package | Raw data, analysis reports, BALF cytology, lung function data, histology slides, ELISA results, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: Why combine cigarette smoke and LPS in the COPD model?
A: Cigarette smoke induces chronic inflammation and emphysema, while LPS (mimicking bacterial infection) exacerbates airway inflammation and mucus production. The combination more closely replicates human COPD pathology, including acute exacerbations.
Q: What are the key similarities with human COPD?
A: The models exhibit progressive airflow obstruction, airway inflammation (macrophages, neutrophils), emphysema, goblet cell metaplasia, and systemic effects, closely resembling human COPD.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different smoke exposure durations, LPS doses)?
A: Absolutely. Our scientific team tailors smoke exposure protocols, LPS dosing, and endpoint analyses to your specific drug candidate.










































