Key Features & Benefits
Clinically relevant – Recapitulates key features of multiple sclerosis pathology: oligodendrocyte loss, demyelination, gliosis, and motor dysfunction.
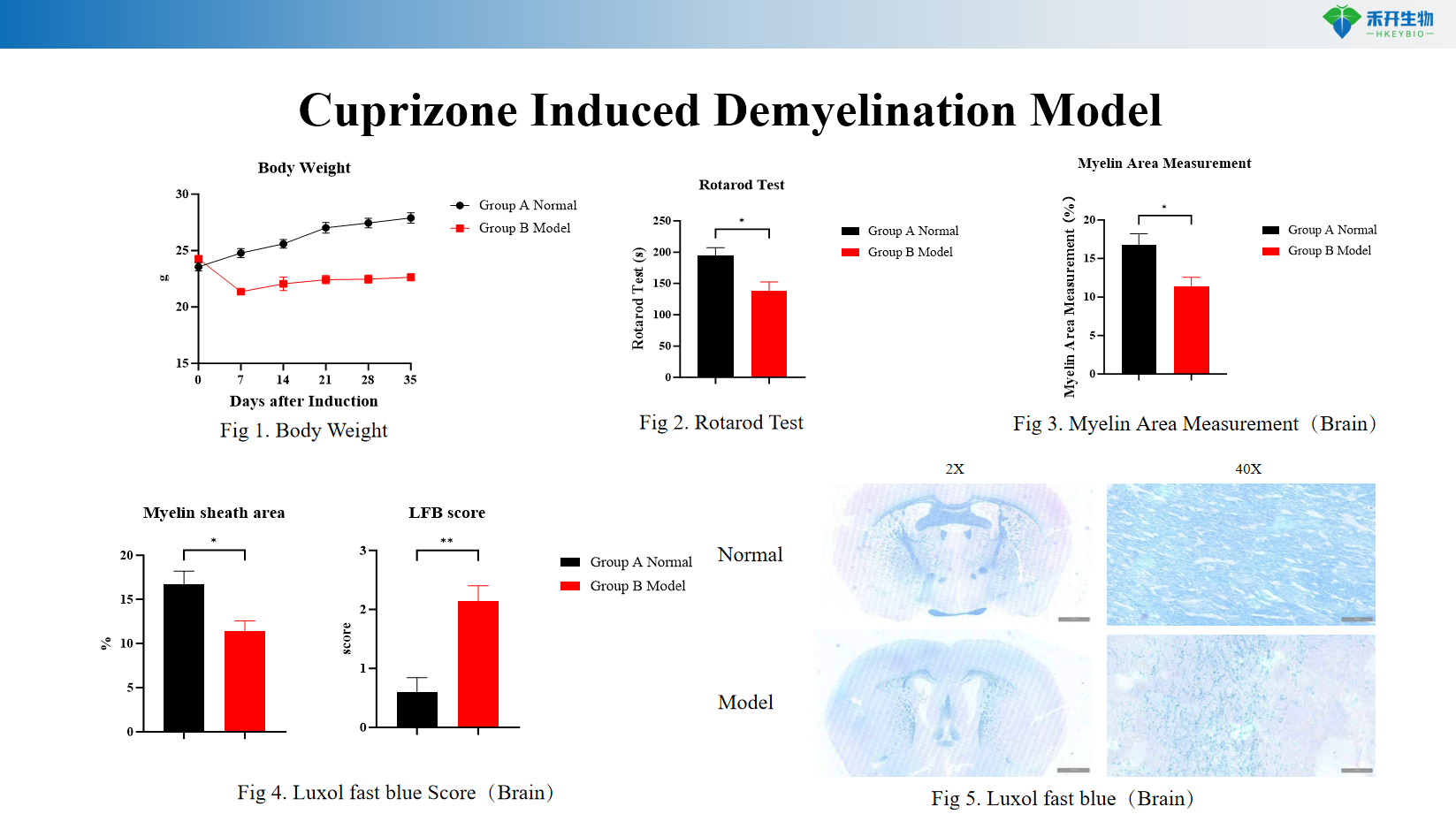
Quantifiable endpoints – Body weight, Rotarod test (motor coordination), myelin area measurement (histology), Luxol fast blue staining and scoring.
Mechanism-driven – Cuprizone induces oligodendrocyte stress through copper chelation, leading to mitochondrial dysfunction and apoptosis, with subsequent glial activation.
Translational value – Ideal for testing remyelinating therapies, neuroprotective agents, and anti-inflammatory drugs for multiple sclerosis and other demyelinating diseases.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Cuprizone Induced Demyelination Model
Applications
• Efficacy testing of remyelinating therapies (anti-LINGO-1, muscarinic receptor antagonists, thyroid hormone agonists)
• Evaluation of neuroprotective agents and anti-inflammatory drugs for multiple sclerosis
• Target validation for oligodendrocyte survival and differentiation pathways
• Biomarker discovery (myelin proteins, glial markers)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | C57BL/6 mouse |
Induction method | Dietary administration of 0.2–0.5% cuprizone mixed in standard rodent chow for 3–6 weeks |
Study duration | 3–8 weeks (demyelination phase) + optional 2–6 weeks (remyelination phase after cuprizone withdrawal) |
Key endpoints | Body weight, Rotarod test (motor coordination), myelin area measurement (histology, corpus callosum), Luxol fast blue staining and scoring, immunohistochemistry for oligodendrocytes (CC1, Olig2), astrocytes (GFAP), microglia (Iba1), optional: electron microscopy for myelin thickness, qPCR for myelin genes (MBP, PLP, MAG) |
Data package | Raw data, analysis reports, behavioral data, histology slides (LFB, IHC), image analysis files, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does cuprizone induce demyelination?
A: Cuprizone is a copper chelator that disrupts mitochondrial function in oligodendrocytes, leading to metabolic stress, oxidative damage, and apoptosis. This triggers microglial activation and phagocytosis of myelin debris, resulting in demyelination, particularly in the corpus callosum.
Q: What are the key similarities with human multiple sclerosis?
A: The model exhibits oligodendrocyte loss, primary demyelination, astrogliosis, microglial activation, and motor function deficits. Unlike EAE, it lacks a significant peripheral immune component, making it ideal for studying central demyelination and remyelination processes.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different cuprizone concentrations, durations, remyelination studies)?
A: Absolutely. Our scientific team tailors cuprizone dosing regimens, study timelines (acute vs. chronic demyelination, remyelination phase), and endpoint analyses to your specific drug candidate.































